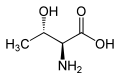
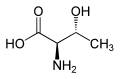
Allothreonine is an amino acid with the formula CH3CH(OH)CH(NH2)CO2H. It is the diastereomer of the amino acid threonine. Like most other amino acids, allothreonine is a water-soluble colorless solid. Although not one of the proteinogenic amino acids, it has often been the subject for the synthesis of novel proteins using an expanded genetic code.[1] Racemic allothreonine can be produced in the laboratory from bromomethoxybutyric acid.[2]
| |||
| Names | |||
|---|---|---|---|
| Other names
(2S,3S)-2-amino-3-hydroxybutanoic acid
| |||
| Identifiers | |||
| |||
3D model (JSmol)
|
| ||
| ChEBI |
| ||
| ChEMBL |
| ||
| ChemSpider | |||
| EC Number |
| ||
| KEGG |
| ||
PubChem CID
|
| ||
| UNII |
| ||
CompTox Dashboard (EPA)
|
| ||
| |||
| |||
| Properties | |||
| C4H9NO3 | |||
| Molar mass | 119.120 g·mol−1 | ||
| Appearance | White solid | ||
| Melting point | 273.5–275.0 °C (524.3–527.0 °F; 546.6–548.1 K) decomposition | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Structure
editThreonine has R, S stereochemistry at carbons 2 and 3 for the naturally occurring stereoisomer and S, R stereochemistry for its enantiomer. Allothreonine has S, S stereochemistry at carbons 2 and 3 in the natural stereoisomer, but R, R in the very rare enantiomer.
| |
| L-Threonine (2S,3R) and D-Threonine (2R,3S) |
| |
| L-Allothreonine (2S,3S) and D-Allothreonine (2R,3R) |
Occurrence
editKatanosins are a group of potent antibiotics contain allothreonine.[3]
Peptides containing the allothreonine residue have also been isolated from natural source.[4]
References
edit- ^ Johnson, Brooke A.; Clark, Kenzie A.; Bushin, Leah B.; Spolar, Calvin N.; Seyedsayamdost, Mohammad R. (2024). "Expanding the Landscape of Noncanonical Amino Acids in RiPP Biosynthesis". Journal of the American Chemical Society. 146 (6): 3805–3815. doi:10.1021/jacs.3c10824. PMID 38316431.
- ^ Carter, Herbert E.; West, Harold D. (1940). "dl-Threonine". Organic Syntheses. 20: 101. doi:10.15227/orgsyn.020.0101.
- ^ Bonner, DP; O'Sullivan, J; Tanaka, SK; Clark, JM; Whitney, RR (1988). "Lysobactin, a Novel Antibacterial Agent Produced by Lysobacter sp. II. Biological Properties". The Journal of Antibiotics. 41 (12): 1745–51. doi:10.7164/antibiotics.41.1745. PMID 3209466.
- ^ Sarabia, Francisco; Chammaa, Samy; García-Ruiz, Cristina (2011). "Solid Phase Synthesis of Globomycin and SF-1902 A5". The Journal of Organic Chemistry. 76 (7): 2132–2144. doi:10.1021/jo1025145. PMID 21366318.