In organometallic chemistry, transition metal complexes of nitrite describes families of coordination complexes containing one or more nitrite (−NO2) ligands.[2] Although the synthetic derivatives are only of scholarly interest, metal-nitrite complexes occur in several enzymes that participate in the nitrogen cycle.[3]
Structure and bonding
editBonding modes
editThree linkage isomers are common for nitrite ligands, O-bonded, N-bonded, and bidentate O,O-bonded. The former two isomers have been characterized for the pentamminecobalt(III) system, i.e. [(NH3)5Co−NO2]2+ and [(NH3)5Co−ONO]2+, referred to as N-nitrito and O-nitrito, respectively. These two forms are sometimes called nitro and nitrito. These isomers can be interconverted in some complexes.[4]
An example of chelating nitrite is [Cu(bipy)2(O2N)]NO3 – "bipy" is the bidentate ligand 2,2′-bipyridyl. This bonding mode is sometimes described as κ2O,O-NO2.
Focusing on electron-counting in monometallic complexes, O-bonded, N-bonded are viewed as 1-electron pseudohalides ("X-ligand"). The bidentate O,O-bonded is an "L-X ligand", akin to bidentate carboxylate.
With respect to HSAB theory, the N bonding mode is more common for softer metal centers. The O and O,O-bidentate modes are hard ligands, being found on Lewis-acidic metal centers.
The kinetically-favored O-bonded isomer [(NH3)5Co−ONO]2+ converts to [(NH3)5Co−NO2]2+. In its reaction with ferric porphyrin complexes, nitrite gives the O-bonded isomer, Fe(porph)ONO. Addition of donor ligands to this complex induces the conversion to the octahedral low-spin isomer, which now is a soft Lewis acid. The nitrite isomerizes to the N-bonded isomer, Fe(porph)NO2(L).[5]
The isomerization of [(NH3)5Co−ONO]2+ to [(NH3)5Co−NO2]2+ proceeds in an intramolecular manner.[6]
Homoleptic complexes
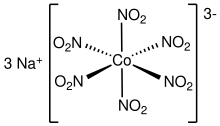
editSeveral homoleptic (complexes with only one kind of ligand) complexes have been characterized by X-ray crystallography. The inventory includes octahedral complexes [M(NO2)6]3−, where M = Co (Sodium cobaltinitrite)[7][8] and Rh. Square-planar homoleptic complexes are also known for Pt(II) and Pd(II). The potassium salts of [M(NO2)4]2− (M = Zn, Cd) feature homoleptic complexes with four O,O-bidentate nitrite ligands.[9]
Synthesis of nitrito complexes
editTraditionally metal nitrito complexes are prepared by salt metathesis or ligand substitution reactions using alkali metal nitrite salts, such as sodium nitrite. At neutral pH, nitrite exists predominantly as the anion, not nitrous acid.[10]
Metal nitrosyl complexes undergo base hydrolysis, yielding nitrite complexes. This pattern is manifested in the behavior of nitroprusside:
- [Fe(CN)5NO]2− + 2 OH− → [Fe(CN)5NO2]4− + H2O
Reactions of nitrito complexes
editSome anionic nitrito complexes undergo acid-induced deoxygenation to give the nitrosyl complex.[11]
- [LnMNO2]− + H+ → [LnMNO]+ + OH−
The reaction is reversible in some cases. Thus, one can generate nitrito complexes by base-hydrolysis of electrophilic metal nitrosyls.
Nitro complexes also catalyze the oxidation of alkenes.[12]
Bioinorganic chemistry
editMetal nitrito complexes figure prominently in the nitrogen cycle, which describes the relationships and interconversions of ammonia up to nitrate. Because nitrogen is often a limiting nutrient, this cycle is important. Nitrite itself does not readily undergo redox reactions, but its metal complexes do.[13]
Oxidation to nitrate
editThe molybdenum-containing enzyme nitrite oxidoreductase catalyzes the oxidation of nitrite to nitrate:
- NO−2 + H2O → NO−3 + 2 H+
Reduction
editThe heme-based enzyme nitrite reductase catalyzes the conversion of nitrite to ammonia. The cycle begins with reduction of an iron-nitrite complex to a metal nitrosyl complex.[3]
The copper-containing enzyme nitrite reductase (CuNIR) catalyzes the 1-electron reduction of nitrite to nitric oxide. The proposed mechanism entails the protonation of a κ2O,O-NO2-Cu(I) complex. This protonation induces cleavage of an N–O bond, giving a HO–Cu–ON center, which features a nitric oxide ligand O-bonded to Cu(II) (an isonitrosyl).
Related compounds
edit- NH4[Co(NO2)4(NH3)2], "Erdmann's salt".[14]
References
edit- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Hitchman, Michael A.; Rowbottom, Graham L. (1982). "Transition Metal Nitrite Complexes". Coordination Chemistry Reviews. 42: 55–132. doi:10.1016/S0010-8545(00)80527-5.
- ^ a b Timmons, A. J.; Symes, M. D. (2015). "Converting between the Oxides of Nitrogen Using Metal–Ligand Coordination Complexes". Chemical Society Reviews. 44 (19): 6708–6722. doi:10.1039/C5CS00269A. PMID 26158348.
- ^ Schaniel, Dominik; Woike, Theo; Behrnd, Norwid-R.; Hauser, Jürg; Krämer, Karl W.; Todorova, Teodora; Delley, Bernard (2009). "Photogeneration of Nitrosyl Linkage Isomers in Octahedrally Coordinated Platinum Complexes in the Red Spectral Range". Inorganic Chemistry. 48 (23): 11399–11406. doi:10.1021/ic901392q. PMID 19863116.
- ^ Ford, Peter C. (2010). "Reactions of NO and Nitrite with Heme Models and Proteins". Inorganic Chemistry. 49 (14): 6226–6239. doi:10.1021/ic902073z. PMID 20666383.
- ^ Jackson, W. Gregory; Lawrance, Geoffrey A.; Lay, Peter A.; Sargeson, Alan M. (1980). "Base-catalyzed nitrito to Nitro Linkage Isomerization of Cobalt(III), Rhodium(III), and Iridium(III) Pentaammine Complexes". Inorganic Chemistry. 19 (4): 904–910. doi:10.1021/ic50206a022.
- ^ Bianchi, R.; Gatti, C.; Adovasio, V.; Nardelli, M. (1996). "Theoretical and experimental (113 K) electron-density study of lithium bis(tetramethylammonium) hexanitrocobaltate(III)". Acta Crystallographica Section B: Structural Science. 52 (3): 471–478. doi:10.1107/S0108768195016879.
- ^ Adovasio, V.; Lina, F.; Nardelli, M.; Pelizzi, G. (1994). "Lithium bis(tetramethyllammonium) Hexanitrocobaltate(III) at 113 and 293 K". Acta Crystallographica Section C: Crystal Structure Communications. 50 (6): 871–874. doi:10.1107/S0108270193011588.
- ^ Ohba, S.; Matsumoto, F.; Takazawa, H.; Saito, Y. (1987). "Structures of cadmium nitrite dihydrate and cadmium dipotassium tetranitrite". Acta Crystallographica Section C: Crystal Structure Communications. 43 (2): 191–194. doi:10.1107/S0108270187096471.
- ^ Harbulak, Edward P.; Albinak, Marvin J. (1966). cis -Dinitrobis(ethylenediamine)cobalt(III) Nitrite and Nitrate. Inorganic Syntheses. pp. 196–198. doi:10.1002/9780470132395.ch51. ISBN 9780470132395.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 449. ISBN 978-0-08-037941-8.
- ^ Fairlamb, Ian J. S. (2015). "Redox-Active NOx Ligands in Palladium-Mediated Processes". Angewandte Chemie International Edition. 54 (36): 10415–10427. doi:10.1002/anie.201411487. PMID 26211412.
- ^ Maia, Luisa B.; Moura, José J. G. (2014). "How Biology Handles Nitrite". Chemical Reviews. 114 (10): 5273–5357. doi:10.1021/cr400518y. PMID 24694090.
- ^ Komiyama, Yoshimichi (1957). "Structures of the Erdmann's Salt, NH4[Co(NH3)2(NO2)4] and Some Other Related Nitro-Ammine-Cobalt (III) Complexes". Bulletin of the Chemical Society of Japan. 30: 13–21. doi:10.1246/bcsj.30.13.