Iodine oxides are chemical compounds of oxygen and iodine. Iodine has only two stable oxides which are isolatable in bulk, iodine tetroxide and iodine pentoxide, but a number of other oxides are formed in trace quantities or have been hypothesized to exist. The chemistry of these compounds is complicated with only a few having been well characterized. Many have been detected in the atmosphere and are believed to be particularly important in the marine boundary layer.[1]
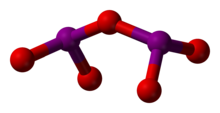
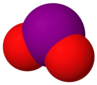
| Molecular formula | I2O | IO[3] | IO2 | I2O4 | I2O5 | I2O6 | I4O9 |
|---|---|---|---|---|---|---|---|
| Name | diiodine oxide | iodine monoxide | iodine dioxide | iodine tetroxide
(diiodine tetroxide) |
Iodine pentoxide
(diiodine pentoxide) |
Diiodine hexaoxide | tetraiodine nonoxide |
| Structure | I2O | IO | IO2 | O2IOIO | O(IO2)2 | [IO2]+[IO4]− | I(OIO2)3 |
| Molecular model | 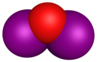
|

|
|

|
|||
| CAS registry | 39319-71-6 | 14696-98-1 | 13494-92-3 | 1024652-24-1 | 12029-98-0 | 65355-99-9 | 66523-94-2 |
| Appearance | Unknown | purple gas | yellow solid | yellow solid | white crystalline solid | yellow solid | dark yellow solid |
| Oxidation state | +1 | +2 | +4 | +3 and +5 | +5 | +5 and +7 | +3 and +5 |
| Melting point | not isolable | not isolable | not isolable | decomp. 100 °C | decomp. 300–350 °C | decomp 150 °C | decomp. 75 °C |
| Specific gravity | 4.2 | 4.8 | 4.53 | ||||
| Solubility in water | decomp. to HIO3 + I2 | 187 g/100 mL | decomp. to HIO3 + I2 |
Diiodine monoxide has largely been the subject of theoretical study,[4] but there is some evidence that it may be prepared in a similar manner to dichlorine monoxide, via a reaction between HgO and I2.[5] The compound appears to be highly unstable but can react with alkenes to give halogenated products.[6]
Radical iodine oxide (IO), iodine dioxide (IO2) and iodine tetroxide ((I2O4) all possess significant and interconnected atmospheric chemistry. They are formed, in very small quantities, at the marine boundary layer by the photooxidation of diiodomethane, which is produced by macroalga such as seaweed or through the oxidation of molecular iodine, produced by the reaction of gaseous ozone and iodide present at the seasurface.[7][8] Despite the small quantities produced (typically below ppt) they are thought to be powerful ozone depletion agents.[9][10]
Diiodine pentoxide (I2O5) is the anhydride of iodic acid and the only stable anhydride of an iodine oxoacid.
Tetraiodine nonoxide (I4O9) has been prepared by the gas-phase reaction of I2 with O3 but has not been extensively studied.[11]
Iodate anions
editIodine oxides also form negatively charged anions, which (associated with complementary cations) are components of acids or salts. These include the iodates and periodates.
Their conjugate acids are:
| Iodine oxidation state | −1 | +1 | +3 | +5 | +7 |
|---|---|---|---|---|---|
| Name | Hydrogen iodide* | Hypoiodous acid | Iodous acid | Iodic acid | Periodic acid |
| Formula | HI | HIO | HIO2 | HIO3 | HIO4 or H5IO6 |
• The -1 oxidation state, hydrogen iodide, is not an oxide, but it is included in this table for completeness.
The periodates include two variants: metaperiodate IO−
4 and orthoperiodate IO5−
6.
See also
editReferences
edit- ^ Kaltsoyannis, Nikolas; Plane, John M. C. (2008). "Quantum chemical calculations on a selection of iodine-containing species (IO, OIO, INO3, (IO)2, I2O3, I2O4 and I2O5) of importance in the atmosphere". Physical Chemistry Chemical Physics. 10 (13): 1723–33. Bibcode:2008PCCP...10.1723K. doi:10.1039/B715687C. PMID 18350176.
- ^ Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ^ Nikitin, I V (31 August 2008). "Halogen monoxides". Russian Chemical Reviews. 77 (8): 739–749. Bibcode:2008RuCRv..77..739N. doi:10.1070/RC2008v077n08ABEH003788. S2CID 250898175.
- ^ Novak, Igor (1998). "Theoretical study of I2O". Heteroatom Chemistry. 9 (4): 383–385. doi:10.1002/(SICI)1098-1071(1998)9:4<383::AID-HC6>3.0.CO;2-9.
- ^ Forbes, Craig P.; Goosen, André; Laue, Hugh A. H. (1974). "Hypoiodite reaction: kinetic study of the reaction of 1,1-diphenyl-ethylene with mercury(II) oxide iodine". Journal of the Chemical Society, Perkin Transactions 1: 2350–2353. doi:10.1039/P19740002350.
- ^ Cambie, Richard C.; Hayward, Rodney C.; Lindsay, Barry G.; Phan, Alice I. T.; Rutledge, Peter S.; Woodgate, Paul D. (1976). "Reactions of iodine oxide with alkenes". Journal of the Chemical Society, Perkin Transactions 1 (18): 1961. doi:10.1039/P19760001961.
- ^ Hoffmann, Thorsten; O'Dowd, Colin D.; Seinfeld, John H. (15 May 2001). "Iodine oxide homogeneous nucleation: An explanation for coastal new particle production" (PDF). Geophysical Research Letters. 28 (10): 1949–1952. Bibcode:2001GeoRL..28.1949H. doi:10.1029/2000GL012399.
- ^ Carpenter, Lucy J.; MacDonald, Samantha M.; Shaw, Marvin D.; Kumar, Ravi; Saunders, Russell W.; Parthipan, Rajendran; Wilson, Julie; Plane, John M.C. (13 January 2013). "Atmospheric iodine levels influenced by seasurface emissions of inorganic iodine" (PDF). Nature Geoscience. 6 (2): 108–111. Bibcode:2013NatGe...6..108C. doi:10.1038/ngeo1687.
- ^ Saiz-Lopez, A.; Fernandez, R. P.; Ordóñez, C.; Kinnison, D. E.; Gómez Martín, J. C.; Lamarque, J.-F.; Tilmes, S. (10 December 2014). "Iodine chemistry in the troposphere and its effect on ozone". Atmospheric Chemistry and Physics. 14 (23): 13119–13143. Bibcode:2014ACP....1413119S. doi:10.5194/acp-14-13119-2014. hdl:11336/100317.
- ^ Cox, R. A.; Bloss, W. J.; Jones, R. L.; Rowley, D. M. (1 July 1999). "OIO and the atmospheric cycle of iodine" (PDF). Geophysical Research Letters. 26 (13): 1857–1860. Bibcode:1999GeoRL..26.1857C. doi:10.1029/1999GL900439. S2CID 128402214.
- ^ Sunder, S.; Wren, J. C.; Vikis, A. C. (December 1985). "Raman spectra of I4O9 formed by the reaction of iodine with ozone". Journal of Raman Spectroscopy. 16 (6): 424–426. Bibcode:1985JRSp...16..424S. doi:10.1002/jrs.1250160611.