Interleukin 11 (IL-11 or adipogenesis inhibitory factor[5]) is a protein that in humans is encoded by the IL11 gene.[6]
IL-11 is a cytokine that was first isolated in 1990 from bone marrow-derived fibrocyte-like stromal cells. It was initially thought to be important for hematopoiesis, notably for megakaryocyte maturation,[7] but subsequently shown to be redundant for platelets, and for other blood cell types, in both mice and humans.[8][9] It was developed as a recombinant protein (rhIL-11) as the drug substance oprelvekin.
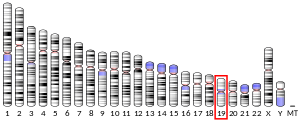
The human IL-11 gene, consisting of 5 exons and 4 introns, is located on chromosome 19,[6] and encodes a 23 kDa protein. IL-11 is a member of the IL-6-type cytokine family, distinguished based on their use of the common co-receptor gp130. Signal specificity is provided by the IL-11Rα subunit which is expressed at high levels in fibroblasts and other stromal cells but not immune cells, unlike IL6 receptors that are expressed at highest levels in immune cells and lowly expressed in stromal cells.[10]
Downstream signalling
editSignal transduction is initiated upon binding of IL-11 to IL-11Rα and gp130, facilitating the formation of higher order structures involving dimers of gp130:Il-11:Il11RA complexes. In some instances, in epithelial-derived cells and cancer cell lines, this permits gp130-associated Janus kinases (JAK) activation and downstream STAT-mediated transcriptional activities.[11] In other instances, in stromal cells, IL-11 activates non-canonical MAPK/ERK-dependent signalling to initiate the post-transcriptional upregulation of specific subsets of transcripts in the absence of an effect on transcription.[10] In fibroblasts, IL-11 drives an ERK-dependent autocrine loop of fibrogenic protein synthesis that is at a nexus of fibrotic signalling and required for the pro-fibrotic activity of TGFB1, PDGF, endothelin1, angiotensin and many other pro-fibrotic factors.[10] IL-11 has also been described in various aspects of tissue regeneration, predominantly in regenerative species like the zebrafish or the axolotl.[12][13] Here, activation of STAT3 by IL-11 is mandatory to allow regeneration and to prevent injury-induced fibrotic remodeling and scar formation.[14]
Function
editIL-11 through its binding to its transmembrane IL-11Rα receptor and resultant activation of downstream signaling pathways has been thought to regulate adipogenesis, osteoclastogenesis, neurogenesis and platelet maturation.[15] More recently it has been discovered that over-expression of IL-11 is associated with a variety of cancers and may provide a link between inflammation and cancer.[15]
IL-11 has been demonstrated to improve platelet recovery after chemotherapy-induced thrombocytopenia, induce acute phase proteins, modulate antigen-antibody responses, participate in the regulation of bone cell proliferation and differentiation IL-11 causes bone-resorption. It stimulates the growth of certain lymphocytes and, in the murine model, stimulates an increase in the cortical thickness and strength of long bones. In addition to having lymphopoietic/hematopoietic and osteotrophic properties, it has functions in many other tissues, including the brain, gut, testis, kidney and bone.[16]
As a signaling molecule, interleukin 11 has a variety of functions associated with its receptor interleukin 11 receptor alpha; such functions include placentation and to some extent decidualization.[17] IL11 has a role during blastocyst implantation in the uteral endometrium; as the blastocyst is imbedded within the endometrium, extravillous trophoblasts invade the maternal spiral arteries for stability and the transfer of life-sustaining elements via the maternal and fetal circulatory systems. This process is highly regulated due to detrimental consequences that can arise from aberrations of the placentation process: poor infiltration of trophoblasts may result in preeclampsia, while severely invasive trophoblasts may resolve in placenta accreta, increta or percreta; all defects that most likely would result in the early demise of the embryo and/or negative effects upon the mother.[17] IL11 is present in the decidua and chorionic villi to regulate the extent in which the placenta implants itself; regulations to ensure maternal well-being and the growth and survival of the fetus. A murine knockout model has been produced for this particular gene, with initial studies involving IL11 role in bone pathologies but have since progressed to fertility research; further research utilizes endometrial and gestational tissue from humans.[17][18]
Manufacture
editInterleukin 11 is manufactured using recombinant DNA technology.[citation needed]
Therapeutic target
editA recombinant form of IL-11, oprelvekin, is a protein therapeutic used for the prevention of severe thrombocytopenia in cancer patients.[19][20]
As IL-11 over-expression is associated with a number of cancers, inhibition of its signaling pathway may have utility in treating cancer,[21] and has also been researched for its potential ability to slow down aging.[22][23]
Transforming growth factor β1 (TGFβ1) through up-regulation of IL-11, stimulates collagen production and is important in wound healing. However dysregulation of TGFβ1 and downstream IL-11 is associated with fibrotic diseases hence inhibition of IL-11 may have utility in treating fibrosis.[10] This cytokine promotes recruitment of immune suppressive cancer-associated fibroblasts to tumors and facilitates chemoresistance.[24]
It is also under investigation as a way to allow diabetes-damaged kidney tissue to regenerate.[25]
See also
editReferences
edit- ^ a b c GRCh38: Ensembl release 89: ENSG00000095752 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000004371 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Kawashima I, Ohsumi J, Mita-Honjo K, Shimoda-Takano K, Ishikawa H, Sakakibara S, et al. (June 1991). "Molecular cloning of cDNA encoding adipogenesis inhibitory factor and identity with interleukin-11". FEBS Letters. 283 (2): 199–202. doi:10.1016/0014-5793(91)80587-S. PMID 1828438. S2CID 1385397.
- ^ a b McKinley D, Wu Q, Yang-Feng T, Yang YC (July 1992). "Genomic sequence and chromosomal location of human interleukin-11 gene (IL11)". Genomics. 13 (3): 814–9. doi:10.1016/0888-7543(92)90158-O. PMID 1386338.
- ^ Paul SR, Bennett F, Calvetti JA, Kelleher K, Wood CR, O'Hara RM, et al. (October 1990). "Molecular cloning of a cDNA encoding interleukin 11, a stromal cell-derived lymphopoietic and hematopoietic cytokine". Proceedings of the National Academy of Sciences of the United States of America. 87 (19): 7512–6. Bibcode:1990PNAS...87.7512P. doi:10.1073/pnas.87.19.7512. PMC 54777. PMID 2145578.
- ^ Nandurkar HH, Robb L, Tarlinton D, Barnett L, Köntgen F, Begley CG (September 1997). "Adult mice with targeted mutation of the interleukin-11 receptor (IL11Ra) display normal hematopoiesis". Blood. 90 (6): 2148–59. doi:10.1182/blood.V90.6.2148. PMID 9310465.
- ^ Brischoux-Boucher E, Trimouille A, Baujat G, Goldenberg A, Schaefer E, Guichard B, et al. (October 2018). "IL11RA-related Crouzon-like autosomal recessive craniosynostosis in 10 new patients: Resemblances and differences". Clinical Genetics. 94 (3–4): 373–380. doi:10.1111/cge.13409. PMID 29926465. S2CID 49339003.
- ^ a b c d Schafer S, Viswanathan S, Widjaja AA, Lim WW, Moreno-Moral A, DeLaughter DM, et al. (December 2017). "IL-11 is a crucial determinant of cardiovascular fibrosis". Nature. 552 (7683): 110–115. Bibcode:2017Natur.552..110S. doi:10.1038/nature24676. hdl:10044/1/54929. PMC 5807082. PMID 29160304.
- ^ Heinrich PC, Behrmann I, Haan S, Hermanns HM, Müller-Newen G, Schaper F (August 2003). "Principles of interleukin (IL)-6-type cytokine signalling and its regulation". The Biochemical Journal. 374 (Pt 1): 1–20. doi:10.1042/BJ20030407. PMC 1223585. PMID 12773095.
- ^ Gerber T, Murawala P, Knapp D, Masselink W, Schuez M, Hermann S, et al. (October 2018). "Single-cell analysis uncovers convergence of cell identities during axolotl limb regeneration". Science. 362 (6413): eaaq0681. Bibcode:2018Sci...362..681G. doi:10.1126/science.aaq0681. PMC 6669047. PMID 30262634.
- ^ Allanki S, Strilic B, Scheinberger L, Onderwater YL, Marks A, Günther S, et al. (September 2021). "Interleukin-11 signaling promotes cellular reprogramming and limits fibrotic scarring during tissue regeneration". Science Advances. 7 (37): eabg6497. Bibcode:2021SciA....7.6497A. doi:10.1126/sciadv.abg6497. PMC 8442930. PMID 34516874.
- ^ Allanki S, Strilic B, Scheinberger L, Onderwater YL, Marks A, Günther S, et al. (September 2021). "Interleukin-11 signaling promotes cellular reprogramming and limits fibrotic scarring during tissue regeneration". Science Advances. 7 (37): eabg6497. Bibcode:2021SciA....7.6497A. doi:10.1126/sciadv.abg6497. PMC 8442930. PMID 34516874.
- ^ a b Xu DH, Zhu Z, Wakefield MR, Xiao H, Bai Q, Fang Y (April 2016). "The role of IL-11 in immunity and cancer". Cancer Letters. 373 (2): 156–63. doi:10.1016/j.canlet.2016.01.004. PMID 26826523.
- ^ Sims NA, Jenkins BJ, Nakamura A, Quinn JM, Li R, Gillespie MT, et al. (July 2005). "Interleukin-11 receptor signaling is required for normal bone remodeling". Journal of Bone and Mineral Research. 20 (7): 1093–102. doi:10.1359/JBMR.050209. PMID 15940362. S2CID 23648686.
- ^ a b c Paiva P, Salamonsen LA, Manuelpillai U, Walker C, Tapia A, Wallace EM, et al. (November 2007). "Interleukin-11 promotes migration, but not proliferation, of human trophoblast cells, implying a role in placentation". Endocrinology. 148 (11): 5566–72. doi:10.1210/en.2007-0517. PMID 17702845.
- ^ Chen HF, Lin CY, Chao KH, Wu MY, Yang YS, Ho HN (May 2002). "Defective production of interleukin-11 by decidua and chorionic villi in human anembryonic pregnancy". The Journal of Clinical Endocrinology and Metabolism. 87 (5): 2320–8. doi:10.1210/jc.87.5.2320. PMID 11994383.
- ^ Sitaraman SV, Gewirtz AT (October 2001). "Oprelvekin. Genetics Institute". Current Opinion in Investigational Drugs. 2 (10): 1395–400. PMID 11890354.
- ^ "Oprelvekin Injection". RxList.
- ^ Putoczki TL, Ernst M (2015). "IL-11 signaling as a therapeutic target for cancer". Immunotherapy. 7 (4): 441–53. doi:10.2217/imt.15.17. PMID 25917632.
- ^ Widjaja AA, Lim WW, Viswanathan S, Chothani S, Corden B, Dasan CM, et al. (July 2024). "Inhibition of IL-11 signalling extends mammalian healthspan and lifespan". Nature. doi:10.1038/s41586-024-07701-9. PMC 11291288. PMID 39020175.
- ^ Ledford H (2024-07-17). "Mice live longer when inflammation-boosting protein is blocked". Nature. 631 (8022): 716–717. doi:10.1038/d41586-024-02298-5.
- ^ Tao L, Huang G, Wang R, Pan Y, He Z, Chu X, et al. (December 2016). "Cancer-associated fibroblasts treated with cisplatin facilitates chemoresistance of lung adenocarcinoma through IL-11/IL-11R/STAT3 signaling pathway". Scientific Reports. 6: 38408. Bibcode:2016NatSR...638408T. doi:10.1038/srep38408. PMC 5138853. PMID 27922075.
- ^ Irving M (2023-02-13). "Scientists regenerate kidneys to reverse diabetes damage in mice". New Atlas. Retrieved 2023-02-13.
Further reading
edit- Yang YC, Yin T (December 1992). "Interleukin-11 and its receptor". BioFactors. 4 (1): 15–21. PMID 1292471.
- Bhatia M, Davenport V, Cairo MS (January 2007). "The role of interleukin-11 to prevent chemotherapy-induced thrombocytopenia in patients with solid tumors, lymphoma, acute myeloid leukemia and bone marrow failure syndromes". Leukemia & Lymphoma. 48 (1): 9–15. doi:10.1080/10428190600909115. PMID 17325843. S2CID 43024459.
- McKinley D, Wu Q, Yang-Feng T, Yang YC (July 1992). "Genomic sequence and chromosomal location of human interleukin-11 gene (IL11)". Genomics. 13 (3): 814–9. doi:10.1016/0888-7543(92)90158-O. PMID 1386338.
- Kawashima I, Ohsumi J, Mita-Honjo K, Shimoda-Takano K, Ishikawa H, Sakakibara S, et al. (June 1991). "Molecular cloning of cDNA encoding adipogenesis inhibitory factor and identity with interleukin-11". FEBS Letters. 283 (2): 199–202. doi:10.1016/0014-5793(91)80587-S. PMID 1828438. S2CID 1385397.
- Paul SR, Bennett F, Calvetti JA, Kelleher K, Wood CR, O'Hara RM, et al. (October 1990). "Molecular cloning of a cDNA encoding interleukin 11, a stromal cell-derived lymphopoietic and hematopoietic cytokine". Proceedings of the National Academy of Sciences of the United States of America. 87 (19): 7512–6. Bibcode:1990PNAS...87.7512P. doi:10.1073/pnas.87.19.7512. PMC 54777. PMID 2145578.
- Wang XY, Fuhrer DK, Marshall MS, Yang YC (November 1995). "Interleukin-11 induces complex formation of Grb2, Fyn, and JAK2 in 3T3L1 cells". The Journal of Biological Chemistry. 270 (47): 27999–8002. doi:10.1074/jbc.270.47.27999. PMID 7499280.
- Chérel M, Sorel M, Lebeau B, Dubois S, Moreau JF, Bataille R, et al. (October 1995). "Molecular cloning of two isoforms of a receptor for the human hematopoietic cytokine interleukin-11". Blood. 86 (7): 2534–40. doi:10.1182/blood.V86.7.2534.2534. PMID 7670098.
- Yamaguchi M, Miki N, Ono M, Ohtsuka C, Demura H, Kurachi H, et al. (March 1995). "Inhibition of growth hormone-releasing factor production in mouse placenta by cytokines using gp130 as a signal transducer". Endocrinology. 136 (3): 1072–8. doi:10.1210/endo.136.3.7867561. PMID 7867561.
- Mehler MF, Rozental R, Dougherty M, Spray DC, Kessler JA (March 1993). "Cytokine regulation of neuronal differentiation of hippocampal progenitor cells". Nature. 362 (6415): 62–5. Bibcode:1993Natur.362...62M. doi:10.1038/362062a0. PMID 8383296. S2CID 4313610.
- Morris JC, Neben S, Bennett F, Finnerty H, Long A, Beier DR, et al. (October 1996). "Molecular cloning and characterization of murine interleukin-11". Experimental Hematology. 24 (12): 1369–76. PMID 8913282.
- Neddermann P, Graziani R, Ciliberto G, Paonessa G (November 1996). "Functional expression of soluble human interleukin-11 (IL-11) receptor alpha and stoichiometry of in vitro IL-11 receptor complexes with gp130". The Journal of Biological Chemistry. 271 (48): 30986–91. doi:10.1074/jbc.271.48.30986. PMID 8940087.
- Barton VA, Hudson KR, Heath JK (February 1999). "Identification of three distinct receptor binding sites of murine interleukin-11". The Journal of Biological Chemistry. 274 (9): 5755–61. doi:10.1074/jbc.274.9.5755. PMID 10026196.
- Tacken I, Dahmen H, Boisteau O, Minvielle S, Jacques Y, Grötzinger J, et al. (October 1999). "Definition of receptor binding sites on human interleukin-11 by molecular modeling-guided mutagenesis". European Journal of Biochemistry. 265 (2): 645–55. doi:10.1046/j.1432-1327.1999.00755.x. PMID 10504396.
- Mahboubi K, Biedermann BC, Carroll JM, Pober JS (April 2000). "IL-11 activates human endothelial cells to resist immune-mediated injury". Journal of Immunology. 164 (7): 3837–46. doi:10.4049/jimmunol.164.7.3837. PMID 10725745.
- Barton VA, Hall MA, Hudson KR, Heath JK (November 2000). "Interleukin-11 signals through the formation of a hexameric receptor complex". The Journal of Biological Chemistry. 275 (46): 36197–203. doi:10.1074/jbc.M004648200. PMID 10948192.
- Curti A, Tafuri A, Ricciardi MR, Tazzari P, Petrucci MT, Fogli M, et al. (April 2002). "Interleukin-11 induces proliferation of human T-cells and its activity is associated with downregulation of p27(kip1)". Haematologica. 87 (4): 373–80. PMID 11940481.
- Van der Meeren A, Mouthon MA, Gaugler MH, Vandamme M, Gourmelon P (June 2002). "Administration of recombinant human IL11 after supralethal radiation exposure promotes survival in mice: interactive effect with thrombopoietin". Radiation Research. 157 (6): 642–9. doi:10.1667/0033-7587(2002)157[0642:AORHIA]2.0.CO;2. PMID 12005542. S2CID 21496463.
- McCloy MP, Roberts IA, Howarth LJ, Watts TL, Murray NA (June 2002). "Interleukin-11 levels in healthy and thrombocytopenic neonates". Pediatric Research. 51 (6): 756–60. doi:10.1203/00006450-200206000-00016. PMID 12032273.
- Bartz H, Büning-Pfaue F, Türkel O, Schauer U (September 2002). "Respiratory syncytial virus induces prostaglandin E2, IL-10 and IL-11 generation in antigen presenting cells". Clinical and Experimental Immunology. 129 (3): 438–45. doi:10.1046/j.1365-2249.2002.01927.x. PMC 1906469. PMID 12197884.