The Barton–Kellogg reaction is a coupling reaction between a diazo compound and a thioketone, giving an alkene by way of an episulfide intermediate.[1][2][3] The Barton–Kellogg reaction is also known as Barton–Kellogg olefination[4] and Barton olefin synthesis.[5]
| Barton–Kellogg reaction | |
|---|---|
| Named after | Sir Derek Barton Richard M. Kellogg |
| Reaction type | Coupling reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000495 |
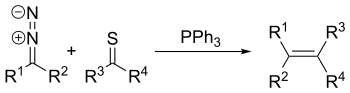
This reaction was pioneered by Hermann Staudinger,[6] and also goes by the name Staudinger type diazo-thioketone coupling.
Reaction mechanism
editIn the reaction mechanism for this reaction, the diazo compound reacts as a 1,3-dipole in a 1,3-dipolar cycloaddition with the thioketone to give a 5-membered thiadiazoline ring. This intermediate is unstable; it extrudes a molecule of nitrogen to form a thiocarbonyl ylide, which then cyclizes to form a stable episulfide. Triphenylphosphine reacts as a nucleophile, opening the three-membered ring to form a sulfaphosphatane. In a manner similar to the Wittig reaction, this structure then expels triphenylphosphine sulfide to produce an alkene.
Scope
editThe diazo compound can be obtained from a ketone by reaction with hydrazine to a hydrazone followed by oxidation. Many reagents exist for this conversion for example silver(I) oxide and (bis(trifluoroacetoxy)iodo)benzene.[7] The thioketone required for this reaction can be obtained from a ketone and phosphorus pentasulfide. Desulfurization of the episulfide can be accomplished by many phosphines and also by copper powder.
The main advantage of this reaction over the McMurry reaction is the notion that the reaction can take place with two different ketones. In this regard the diazo-thioketone coupling is a cross-coupling rather than a homocoupling.
References
edit- ^ D. H. R. Barton & B. J. Willis (1970). "Olefin synthesis by twofold extrusion processes". J. Chem. Soc. D (19): 1225. doi:10.1039/C29700001225.
- ^ R. M. Kellogg & S. Wassenaar (1970). "Thiocarbonyl ylides. An approach to "tetravalent sulfur" compounds". Tetrahedron Lett. 11 (23): 1987. doi:10.1016/S0040-4039(01)98134-1.
- ^ R. M. Kellogg (1976). "The molecules R2CXCR2 including azomethine, carbonyl and thiocarbonyl ylides. Their syntheses, properties and reactions". Tetrahedron. 32 (18): 2165–2184. doi:10.1016/0040-4020(76)85131-9.
- ^ "Barton-Kellogg olefination". Comprehensive Organic Name Reactions and Reagents. 2010. pp. 249–253. doi:10.1002/9780470638859.conrr056. ISBN 9780470638859.
- ^ "Barton olefin synthesis". Merck Index (15th ed.).
- ^ H. Staudinger & J. Siegwart (1920). "Einwirkungen von aliphatischen Diazoverbindungen auf Thioketone". Helv. Chim. Acta. 3: 833–840. doi:10.1002/hlca.19200030178.
- ^ Matthijs K. J. ter Wiel; Javier Vicario; Stephen G. Davey; Auke Meetsma & Ben L. Feringa (2005). "New procedure for the preparation of highly sterically hindered alkenes using a hypervalent iodine reagent" (PDF). Organic & Biomolecular Chemistry. 3 (1): 28–30. doi:10.1039/b414959a. PMID 15602594.