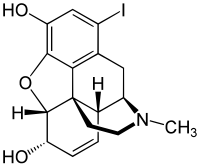
1-Iodomorphine is a semi-synthetic narcotic analgesic formed by halogenation of the 1 position on the morphine carbon skeleton.[1][2][3] Halogenated morphine derivatives were first synthesised in Germany, Austria/Austria-Hungary, the United Kingdom and the United States in the period 1890 to 1930. Use of this drug increased after 1945 for the below-mentioned research. It is a research chemical which is often prepared in the laboratory when it is needed.
| |
| |
| Names | |
|---|---|
| IUPAC name
1-Iodo-17-methyl-7,8-didehydro-4,5α-epoxymorphinan-3,6α-diol
| |
| Systematic IUPAC name
(4R,4aR,7S,7aR,12bS)-11-Iodo-3-methyl-2,3,4,4a,7,7a-hexahydro-1H-4,12-methano[1]benzofuro[3,2-e]isoquinoline-7,9-diol | |
| Other names
(5α,6α)-7,8-Didehydro-4,5-epoxy-1-iodo-17-methyl-morphinan-3,6-diol
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C17H18INO3 | |
| Molar mass | 411.239 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Along with the similar 2-iodomorphine as well as iodinated analogs of dihydromorphine, dihydrocodeine, heroin, and the fluorinated, chlorinated, and brominated analogues of this series, this change may not impact the activity of the drug to a notable extent[citation needed] but 1- and 2-iodomorphine are used in pharmacological, neurological, metabolic, and endocrine research as it allows the tagging of morphine with iodine-131 or iodine-129.[4] Such research was important in the discovery of opioid receptors in the central nervous system, peripheral nervous system, and other tissues in humans, mammals, birds, and some reptiles, amphibians, fish, insects, and arthropods.
References
edit- ^ Liebman, A. A.; Malarek, D. H.; Blount, J. F.; Nelson, N. R.; Delaney, C. M. (1978). "Preparation and crystal structure of 6-acetyl-1-iodocodeine". The Journal of Organic Chemistry. 43 (4): 737–739. doi:10.1021/jo00398a049.
- ^ Seltzman, H. H.; Roche, M. J.; Laudeman, C. P.; Wyrick, C. D.; Carroll, F. I. (1998). "Protection of the allylic alcohol double bond from catalytic reduction in the preparation of [1-3H]morphine and [1-3H]codeine". Journal of Labelled Compounds and Radiopharmaceuticals. 41 (9): 811–821. doi:10.1002/(SICI)1099-1344(1998090)41:9<811::AID-JLCR132>3.0.CO;2-3.
- ^ Wentland, M. P.; Duan, W.; Cohen, D. J.; Bidlack, J. M. (2000). "Selective Protection and Functionalization of Morphine: Synthesis and Opioid Receptor Binding Properties of 3-Amino-3-desoxymorphine Derivatives". Journal of Medicinal Chemistry. 43 (19): 3558–3565. doi:10.1021/jm000119i. PMID 11000010.
- ^ Tafani, J. A.; Lazorthes, Y.; Danet, B.; Verdie, J. C.; Esquerre, J. P.; Simon, J.; Guiraud, R. (1989). "Human brain and spinal cord scan after intracerebroventricular administration of iodine-123 morphine". International Journal of Radiation Applications and Instrumentation. Part B, Nuclear Medicine and Biology. 16 (5): 505–509. doi:10.1016/0883-2897(89)90064-0. PMID 2807956.