Monoclonal antibodies (mAbs) have varied therapeutic uses. It is possible to create a mAb that binds specifically to almost any extracellular target, such as cell surface proteins and cytokines. They can be used to render their target ineffective (e.g. by preventing receptor binding),[1] to induce a specific cell signal (by activating receptors),[1] to cause the immune system to attack specific cells, or to bring a drug to a specific cell type (such as with radioimmunotherapy which delivers cytotoxic radiation).
Major applications include cancer, autoimmune diseases, asthma, organ transplants, blood clot prevention, and certain infections.
Antibody structure and function
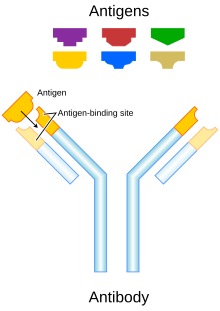
editImmunoglobulin G (IgG) antibodies are large heterodimeric molecules, approximately 150 kDa and are composed of two kinds of polypeptide chain, called the heavy (~50kDa) and the light chain (~25kDa). The two types of light chains are kappa (κ) and lambda (λ). By cleavage with enzyme papain, the Fab (fragment-antigen binding) part can be separated from the Fc (fragment crystallizable region) part of the molecule. The Fab fragments contain the variable domains, which consist of three antibody hypervariable amino acid domains responsible for the antibody specificity embedded into constant regions. The four known IgG subclasses are involved in antibody-dependent cellular cytotoxicity.[2] Antibodies are a key component of the adaptive immune response, playing a central role in both in the recognition of foreign antigens and the stimulation of an immune response to them. The advent of monoclonal antibody technology has made it possible to raise antibodies against specific antigens presented on the surfaces of tumors.[3] Monoclonal antibodies can be acquired in the immune system via passive immunity or active immunity. The advantage of active monoclonal antibody therapy is the fact that the immune system will produce antibodies long-term, with only a short-term drug administration to induce this response. However, the immune response to certain antigens may be inadequate, especially in the elderly. Additionally, adverse reactions from these antibodies may occur because of long-lasting response to antigens.[4] Passive monoclonal antibody therapy can ensure consistent antibody concentration, and can control for adverse reactions by stopping administration. However, the repeated administration and consequent higher cost for this therapy are major disadvantages.[4]
Monoclonal antibody therapy may prove to be beneficial for cancer, autoimmune diseases, and neurological disorders that result in the degeneration of body cells, such as Alzheimer's disease. Monoclonal antibody therapy can aid the immune system because the innate immune system responds to the environmental factors it encounters by discriminating against foreign cells from cells of the body. Therefore, tumor cells that are proliferating at high rates, or body cells that are dying which subsequently cause physiological problems are generally not specifically targeted by the immune system, since tumor cells are the patient's own cells. Tumor cells, however are highly abnormal, and many display unusual antigens. Some such tumor antigens are inappropriate for the cell type or its environment. Monoclonal antibodies can target tumor cells or abnormal cells in the body that are recognized as body cells, but are debilitating to one's health.[citation needed]
History
editImmunotherapy developed in the 1970s following the discovery of the structure of antibodies and the development of hybridoma technology, which provided the first reliable source of monoclonal antibodies.[6][7] These advances allowed for the specific targeting of tumors both in vitro and in vivo. Initial research on malignant neoplasms found mAb therapy of limited and generally short-lived success with blood malignancies.[8][9] Treatment also had to be tailored to each individual patient, which was impracticable in routine clinical settings.[citation needed]
Four major antibody types that have been developed are murine, chimeric, humanised and human. Antibodies of each type are distinguished by suffixes on their name.[citation needed]
Murine
editInitial therapeutic antibodies were murine analogues (suffix -omab). These antibodies have: a short half-life in vivo (due to immune complex formation), limited penetration into tumour sites and inadequately recruit host effector functions.[10] Chimeric and humanized antibodies have generally replaced them in therapeutic antibody applications.[11] Understanding of proteomics has proven essential in identifying novel tumour targets.[citation needed]
Initially, murine antibodies were obtained by hybridoma technology, for which Jerne, Köhler and Milstein received a Nobel prize. However the dissimilarity between murine and human immune systems led to the clinical failure of these antibodies, except in some specific circumstances. Major problems associated with murine antibodies included reduced stimulation of cytotoxicity and the formation of complexes after repeated administration, which resulted in mild allergic reactions and sometimes anaphylactic shock.[10] Hybridoma technology has been replaced by recombinant DNA technology, transgenic mice and phage display.[11]
Chimeric and humanized
editTo reduce murine antibody immunogenicity (attacks by the immune system against the antibody), murine molecules were engineered to remove immunogenic content and to increase immunologic efficiency.[10] This was initially achieved by the production of chimeric (suffix -ximab) and humanized antibodies (suffix -zumab). Chimeric antibodies are composed of murine variable regions fused onto human constant regions. Taking human gene sequences from the kappa light chain and the IgG1 heavy chain results in antibodies that are approximately 65% human. This reduces immunogenicity, and thus increases serum half-life.[citation needed]
Humanised antibodies are produced by grafting murine hypervariable regions on amino acid domains into human antibodies. This results in a molecule of approximately 95% human origin. Humanised antibodies bind antigen much more weakly than the parent murine monoclonal antibody, with reported decreases in affinity of up to several hundredfold.[12][13] Increases in antibody-antigen binding strength have been achieved by introducing mutations into the complementarity determining regions (CDR),[14] using techniques such as chain-shuffling, randomization of complementarity-determining regions and antibodies with mutations within the variable regions induced by error-prone PCR, E. coli mutator strains and site-specific mutagenesis.[15]
Human monoclonal antibodies
editHuman monoclonal antibodies (suffix -umab) are produced using transgenic mice or phage display libraries by transferring human immunoglobulin genes into the murine genome and vaccinating the transgenic mouse against the desired antigen, leading to the production of appropriate monoclonal antibodies.[11] Murine antibodies in vitro are thereby transformed into fully human antibodies.[3]
The heavy and light chains of human IgG proteins are expressed in structural polymorphic (allotypic) forms. Human IgG allotype is one of the many factors that can contribute to immunogenicity.[16][17]
Targeted conditions
editCancer
editAnti-cancer monoclonal antibodies can be targeted against malignant cells by several mechanisms. Ramucirumab is a recombinant human monoclonal antibody and is used in the treatment of advanced malignancies.[18] In childhood lymphoma, phase I and II studies have found a positive effect of using antibody therapy.[19]
Monoclonal antibodies used to boost an anticancer immune response is another strategy to fight cancer where cancer cells are not targeted directly. Strategies include antibodies engineered to block mechanisms which downregulate anticancer immune responses, checkpoints such as PD-1 and CTLA-4 (checkpoint therapy),[20] and antibodies modified to stimulate activation of immune cells.[21]
Autoimmune diseases
editMonoclonal antibodies used for autoimmune diseases include infliximab and adalimumab, which are effective in rheumatoid arthritis, Crohn's disease and ulcerative colitis by their ability to bind to and inhibit TNF-α.[22] Basiliximab and daclizumab inhibit IL-2 on activated T cells and thereby help preventing acute rejection of kidney transplants.[22] Omalizumab inhibits human immunoglobulin E (IgE) and is useful in moderate-to-severe allergic asthma.[citation needed]
Alzheimer's disease
editAlzheimer's disease (AD) is a multi-faceted, age-dependent, progressive neurodegenerative disorder, and is a major cause of dementia.[23] According to the Amyloid hypothesis, the accumulation of extracellular amyloid beta peptides (Aβ) into plaques via oligomerization leads to hallmark symptomatic conditions of AD through synaptic dysfunction and neurodegeneration.[24] Immunotherapy via exogenous monoclonal antibody (mAb) administration has been known to treat various central nervous disorders. In the case of AD, immunotherapy is believed to inhibit Aβ-oligomerization or clearing of Aβ from the brain and thereby prevent neurotoxicity.[25]
However, mAbs are large molecules and due to the blood–brain barrier, uptake of mAb into the brain is extremely limited, only approximately 1 of 1000 mAb molecules is estimated to pass.[25] However, the Peripheral Sink hypothesis proposes a mechanism where mAbs may not need to cross the blood–brain barrier.[26] Therefore, many research studies are being conducted from failed attempts to treat AD in the past.[24]
However, anti-Aβ vaccines can promote antibody-mediated clearance of Aβ plaques in transgenic mice models with amyloid precursor proteins (APP), and can reduce cognitive impairments.[23] Vaccines can stimulate the immune system to produce its own antibodies, in the case of Alzheimer's disease by administration of the antigen Aβ. [27] This is also known as active immunotherapy. Another strategy is so called passive immunotherapy. In this case the antibodies is produced externally in cultured cells and are delivered to the patient in the form of a drug. In mice expressing APP, both active and passive immunization of anti-Aβ antibodies has been shown to be effective in clearing plaques, and can improve cognitive function.[24]
Currently, there are two FDA approved antibody therapies for Alzheimer's disease, Aducanemab and Lecanemab. Aducanemab has received accelerated approval while Lecanemab has received full approval.[25] Several clinical trials using passive and active immunization have been performed and some are on the way with expected results in a couple of years.[24][25] The implementation of these drugs is often during the early onset of AD. Other research and drug development for early intervention and AD prevention is ongoing. Examples of important mAb drugs that have been or are under evaluation for treatment of AD include Bapineuzumab, Solanezumab, Gautenerumab, Crenezumab, Aducanemab, Lecanemab and Donanemab.[25]
Bapineuzumab
editBapineuzumab, a humanized anti-Aβ mAb, is directed against the N-terminus of Aβ. Phase II clinical trials of Bapineuzumab in mild to moderate AD patients resulted in reduced Aβ concentration in the brain. However, in patients with increased apolipoprotein (APOE) e4 carriers, Bapineuzumab treatment is also accompanied by vasogenic edema,[28] a cytotoxic condition where the blood brain barrier has been disrupted thereby affecting white matter from excess accumulation of fluid from capillaries in intracellular and extracellular spaces of the brain.[29]
In Phase III clinical trials, Bapineuzumab showed promising positive effect on biomarkers of AD but failed to show effect on cognitive decline. Therefore, Bapineuzumab was discontinued after failing in the Phase III clinical trial.[29]
Solanezumab
editSolanezumab, an anti-Aβ mAb, targets the N-terminus of Aβ. In Phase I and Phase II of clinical trials, Solanezumab treatment resulted in cerebrospinal fluid elevation of Aβ, thereby showing a reduced concentration of Aβ plaques. Additionally, there are no associated adverse side effects. Phase III clinical trials of Solanezumab brought about significant reduction in cognitive impairment in patients with mild AD, but not in patients with severe AD. However, Aβ concentration did not significantly change, along with other AD biomarkers, including phospho-tau expression, and hippocampal volume. Phase III clinical trials of Solanezumab failed as it did not show effect on cognitive decline in comparison to placebo. [30]
Lecanemab
editLecanemab (BAN2401), is a humanized mAb that selectively targets toxic soluble Aβ protofibrils,[31] In phase 3 clinical trials,[32] Lecanemab showed a 27% slower cognitive decline after 18 months of treatment in comparison to placebo.[33][34] The phase 3 clinical trials also reported infusion related reactions, amyloid-related imaging abnormalities and headaches as the most common side effects of Lecanemab. In July 2023 the FDA gave Lecanemab full approval for the treatment of Alzheimer's Disease [35] and it was given the commercial name Leqembi.
Preventive trials
editFailure of several drugs in Phase III clinical trials has led to AD prevention and early intervention for onset AD treatment endeavours. Passive anti-Aβ mAb treatment can be used for preventive attempts to modify AD progression before it causes extensive brain damage and symptoms. Trials using mAb treatment for patients positive for genetic risk factors, and elderly patients positive for indicators of AD are underway. This includes anti-AB treatment in Asymptomatic Alzheimer's Disease (A4), the Alzheimer's Prevention Initiative (API), and DIAN-TU.[26] The A4 study on older individuals who are positive for indicators of AD but are negative for genetic risk factors will test Solanezumab in Phase III Clinical Trials, as a follow-up of previous Solanezumab studies.[26] DIAN-TU, launched in December 2012, focuses on young patients positive for genetic mutations that are risks for AD. This study uses Solanezumab and Gautenerumab. Gautenerumab, the first fully human MAB that preferentially interacts with oligomerized Aβ plaques in the brain, caused significant reduction in Aβ concentration in Phase I clinical trials, preventing plaque formation and concentration without altering plasma concentration of the brain. Phase II and III clinical trials are currently being conducted.[26]
Therapy types
editRadioimmunotherapy
editRadioimmunotherapy (RIT) involves the use of radioactively-conjugated murine antibodies against cellular antigens. Most research involves their application to lymphomas, as these are highly radio-sensitive malignancies. To limit radiation exposure, murine antibodies were chosen, as their high immunogenicity promotes rapid tumor clearance. Tositumomab is an example used for non-Hodgkin's lymphoma.[citation needed]
Antibody-directed enzyme prodrug therapy
editAntibody-directed enzyme prodrug therapy (ADEPT) involves the application of cancer-associated monoclonal antibodies that are linked to a drug-activating enzyme. Systemic administration of a non-toxic agent results in the antibody's conversion to a toxic drug, resulting in a cytotoxic effect that can be targeted at malignant cells. The clinical success of ADEPT treatments is limited.[36]
Antibody-drug conjugates
editAntibody-drug conjugates (ADCs) are antibodies linked to one or more drug molecules. Typically when the ADC meets the target cell (e.g. a cancerous cell) the drug is released to kill it. Many ADCs are in clinical development. As of 2016[update] a few have been approved.[citation needed]
Immunoliposome therapy
editImmunoliposomes are antibody-conjugated liposomes. Liposomes can carry drugs or therapeutic nucleotides and when conjugated with monoclonal antibodies, may be directed against malignant cells. Immunoliposomes have been successfully used in vivo to convey tumour-suppressing genes into tumours, using an antibody fragment against the human transferrin receptor. Tissue-specific gene delivery using immunoliposomes has been achieved in brain and breast cancer tissue.[37]
Checkpoint therapy
editCheckpoint therapy uses antibodies and other techniques to circumvent the defenses that tumors use to suppress the immune system. Each defense is known as a checkpoint. Compound therapies combine antibodies to suppress multiple defensive layers. Known checkpoints include CTLA-4 targeted by ipilimumab, PD-1 targeted by nivolumab and pembrolizumab and the tumor microenvironment.[20]
The tumor microenvironment (TME) features prevents the recruitment of T cells to the tumor. Ways include chemokine CCL2 nitration, which traps T cells in the stroma. Tumor vasculature helps tumors preferentially recruit other immune cells over T cells, in part through endothelial cell (EC)–specific expression of FasL, ETBR, and B7H3. Myelomonocytic and tumor cells can up-regulate expression of PD-L1, partly driven by hypoxic conditions and cytokine production, such as IFNβ. Aberrant metabolite production in the TME, such as the pathway regulation by IDO, can affect T cell functions directly and indirectly via cells such as Treg cells. CD8 cells can be suppressed by B cells regulation of TAM phenotypes. Cancer-associated fibroblasts (CAFs) have multiple TME functions, in part through extracellular matrix (ECM)–mediated T cell trapping and CXCL12-regulated T cell exclusion.[38]
FDA-approved therapeutic antibodies
editThis section needs to be updated. (May 2021) |
The first FDA-approved therapeutic monoclonal antibody was a murine IgG2a CD3 specific transplant rejection drug, OKT3 (also called muromonab), in 1986. This drug found use in solid organ transplant recipients who became steroid resistant.[39] Hundreds of therapies are undergoing clinical trials. Most are concerned with immunological and oncological targets.
| Antibody | Brand name | Company | Approval date | Route | Type | Target | Indication (Targeted disease) |
BLA STN | Drug Label |
|---|---|---|---|---|---|---|---|---|---|
| abciximab | ReoPro | Centocor | 12/22/1994 | intravenous | chimeric Fab | GPIIb/IIIa | Percutaneous coronary intervention | 103575 | Link |
| adalimumab | Humira | Abbvie | 12/31/2002 | subcutaneous | fully human | TNF | Rheumatoid arthritis | 125057 | Link |
| adalimumab-adbm | Cyltezo | Boehringer Ingelheim | 8/25/17 | subcutaneous | fully human, biosimilar | TNF | Rheumatoid arthritis Juvenile idiopathic arthritis Psoriatic arthritis Ankylosing spondylitis Crohn's disease Ulcerative colitis Plaque psoriasis |
761058 | Link |
| adalimumab-atto | Amjevita | Amgen | 9/23/2016 | subcutaneous | fully human, biosimilar | TNF | Rheumatoid arthritis Juvenile idiopathic arthritis Psoriatic arthritis Ankylosing spondylitis Crohn's disease Ulcerative colitis Plaque psoriasis |
761024 | Link |
| ado-trastuzumab emtansine | Kadcyla | Genentech | 2/22/2013 | intravenous | humanized, antibody-drug conjugate | HER2 | Metastatic breast cancer | 125427 | Link |
| alemtuzumab | Campath, Lemtrada | Genzyme | 5/7/2001 | intravenous | humanized | CD52 | B-cell chronic lymphocytic leukemia | 103948 | Link |
| alirocumab | Praluent | Sanofi Aventis | 7/24/2015 | subcutaneous | fully human | PCSK9 | Heterozygous familial hypercholesterolemia Refractory hypercholesterolemia |
125559 | Link |
| atezolizumab | Tecentriq | Genentech | 5/18/2016 | intravenous | humanized | PD-L1 | Urothelial carcinoma | 761034 | Link |
| atezolizumab | Tecentriq | Genentech | 10/18/2016 | intravenous | humanized | PD-L1 | Urothelial carcinoma Metastatic non-small cell lung cancer |
761041 | Link |
| avelumab | Bavencio | EMD Serono | 3/23/2017 | intravenous | fully human | PD-L1 | Metastatic Merkel cell carcinoma | 761049 | Link |
| basiliximab | Simulect | Novartis | 5/12/1998 | intravenous | chimeric | IL2RA | Prophylaxis of acute organ rejection in renal transplant | 103764 | Link |
| belimumab | Benlysta | Human Genome Sciences | 3/9/2011 | intravenous | fully human | BLyS | Systemic lupus erythematosus | 125370 | Link |
| benralizumab | Fasenra | AstraZeneca | 11/14/17 | subcutaneous | humanized | interleukin-5 receptor alpha subunit | Severe asthma, eosinophilic phenotype | 761070 | Link |
| bevacizumab | Avastin | Genentech | 2/26/2004 | intravenous | humanized | VEGF | Metastatic colorectal cancer | 125085 | Link |
| bevacizumab-awwb | Mvasi | Amgen | 9/14/17 | intravenous | humanized, biosimilar | VEGF | Metastatic colorectal cancer Non-squamous Non-small-cell lung carcinoma Glioblastoma Metastatic renal cell carcinoma Cervical cancer |
761028 | Link |
| bezlotoxumab | Zinplava | Merck | 10/21/2016 | intravenous | fully human | Clostridioides difficile toxin B | Prevent recurrence of Clostridioides difficile infection | 761046 | Link |
| blinatumomab | Blincyto | Amgen | 12/3/2014 | intravenous | mouse, bispecific | CD19 | Precursor B-cell acute lymphoblastic leukemia | 125557 | Link |
| brentuximab vedotin | Adcetris | Seattle Genetics | 9/19/2011 | intravenous | chimeric, antibody-drug conjugate | CD30 | Hodgkin lymphoma Anaplastic large-cell lymphoma |
125388 | Link |
| brodalumab | Siliq | Valeant | 2/15/2017 | subcutaneous | chimeric | IL17RA | Plaque psoriasis | 761032 | Link |
| burosumab-twza | Crysvita | Ultragenyx | 4/17/18 | subcutaneous | fully human | FGF23 | X-linked hypophosphatemia | 761068 | Link |
| canakinumab | Ilaris | Novartis | 6/17/2009 | subcutaneous | fully human | IL1B | Cryopyrin-associated periodic syndrome | 125319 | Link |
| capromab pendetide | ProstaScint | Cytogen | 10/28/1996 | intravenous | murine, radiolabeled | PSMA | Diagnostic imaging agent in newly diagnosed prostate cancer or post-prostatectomy | 103608 | Link |
| certolizumab pegol | Cimzia | UCB (company) | 4/22/2008 | subcutaneous | humanized | TNF | Crohn's disease | 125160 | Link |
| cetuximab | Erbitux | ImClone Systems | 2/12/2004 | intravenous | chimeric | EGFR | Metastatic colorectal carcinoma | 125084 | Link |
| daclizumab | Zenapax | Roche | 12/10/1997 | intravenous | humanized | IL2RA | Prophylaxis of acute organ rejection in renal transplant | 103749 | Link |
| daclizumab | Zinbryta | Biogen | 5/27/2016 | subcutaneous | humanized | IL2R | Multiple sclerosis | 761029 | Link |
| daratumumab | Darzalex | Janssen Biotech | 11/16/2015 | intravenous | fully human | CD38 | Multiple myeloma | 761036 | Link |
| denosumab | Prolia, Xgeva | Amgen | 6/1/2010 | subcutaneous | fully human | RANKL | Postmenopausal women with osteoporosis | 125320 | Link |
| dinutuximab | Unituxin | United Therapeutics | 3/10/2015 | intravenous | chimeric | GD2 | Pediatric high-risk neuroblastoma | 125516 | Link |
| dupilumab | Dupixent | Regeneron Pharmaceuticals | 3/28/2017 | subcutaneous | fully human | IL4RA | Atopic dermatitis, asthma | 761055 | Link |
| durvalumab | Imfinzi | AstraZeneca | 5/1/2017 | intravenous | fully human | PD-L1 | Urothelial carcinoma | 761069 | Link |
| eculizumab | Soliris | Alexion | 3/16/2007 | intravenous | humanized | Complement component 5 | Paroxysmal nocturnal hemoglobinuria | 125166 | Link |
| elotuzumab | Empliciti | Bristol-Myers Squibb | 11/30/2015 | intravenous | humanized | SLAMF7 | Multiple myeloma | 761035 | Link |
| emicizumab-kxwh | Hemlibra | Genentech | 11/16/17 | subcutaneous | humanized, bispecific | Factor IXa, Factor X | Hemophilia A (congenital Factor VIII deficiency) with Factor VIII inhibitors. | 761083 | Link |
| erenumab-aooe | Aimovig | Amgen | 5/17/18 | subcutaneous | fully human | CGRP receptor | Migraine headache prevention | 761077 | Link |
| evolocumab | Repatha | Amgen | 8/27/2015 | subcutaneous | fully human | PCSK9 | Heterozygous familial hypercholesterolemia Refractory hypercholesterolemia |
125522 | Link |
| gemtuzumab ozogamicin | Mylotarg | Wyeth | 9/1/17 | intravenous | humanized, antibody-drug conjugate | CD33 | Acute myeloid leukemia | 761060 | Link |
| golimumab | Simponi | Centocor | 4/24/2009 | subcutaneous | fully human | TNF | Rheumatoid arthritis Psoriatic arthritis Ankylosing spondylitis |
125289 | Link |
| golimumab | Simponi Aria | Janssen Biotech | 7/18/2013 | intravenous | fully human | TNF | Rheumatoid arthritis | 125433 | Link |
| guselkumab | Tremfya | Janssen Biotech | 7/13/17 | subcutaneous | fully human | IL23 | Plaque psoriasis | 761061 | Link |
| ibalizumab-uiyk | Trogarzo | TaiMed Biologics | 3/6/18 | intravenous | humanized | CD4 | HIV | 761065 | Link |
| ibritumomab tiuxetan | Zevalin | Spectrum Pharmaceuticals | 2/19/2002 | intravenous | murine, radioimmunotherapy | CD20 | Relapsed or refractory low-grade, follicular, or transformed B-cell non-Hodgkin's lymphoma | 125019 | Link |
| idarucizumab | Praxbind | Boehringer Ingelheim | 10/16/2015 | intravenous | humanized Fab | dabigatran | Emergency reversal of anticoagulant dabigatran | 761025 | Link |
| infliximab | Remicade | Centocor | 8/24/1998 | intravenous | chimeric | TNF alpha | Crohn's disease | 103772 | Link |
| infliximab-abda | Renflexis | Samsung Bioepis | 4/21/2017 | intravenous | chimeric, biosimilar | TNF | Crohn's disease Ulcerative colitis Rheumatoid arthritis Ankylosing spondylitis Psoriatic arthritis Plaque psoriasis |
761054 | Link |
| infliximab-dyyb | Inflectra | Celltrion Healthcare | 4/5/2016 | intravenous | chimeric, biosimilar | TNF | Crohn's disease Ulcerative colitis Rheumatoid arthritis Ankylosing spondylitis Psoriatic arthritis Plaque psoriasis |
125544 | Link |
| infliximab-qbtx | Ixifi | Pfizer | 12/13/17 | intravenous | chimeric, biosimilar | TNF | Crohn's disease Ulcerative colitis Rheumatoid arthritis Ankylosing spondylitis Psoriatic arthritis Plaque psoriasis |
761072 | Link |
| inotuzumab ozogamicin | Besponsa | Wyeth | 8/17/17 | intravenous | humanized, antibody-drug conjugate | CD22 | Precursor B-cell acute lymphoblastic leukemia | 761040 | Link |
| ipilimumab | Yervoy | Bristol-Myers Squibb | 3/25/2011 | intravenous | fully human | CTLA-4 | Metastatic melanoma | 125377 | Link |
| ixekizumab | Taltz | Eli Lilly | 3/22/2016 | subcutaneous | humanized | IL17A | Plaque psoriasis | 125521 | Link |
| marstacimab | Hympavzi | Pfizer | 10/11/2024 | intravenous | fully human | Tissue factor pathway inhibitor | Hemophilia A and B | 761345 | [1] |
| mepolizumab | Nucala | GlaxoSmithKline | 11/4/2015 | subcutaneous | humanized | IL5 | Severe asthma | 125526 | Link |
| natalizumab | Tysabri | Biogen Idec | 11/23/2004 | intravenous | humanized | alpha-4 integrin | Multiple sclerosis | 125104 | Link |
| necitumumab | Portrazza | Eli Lilly | 11/24/2015 | intravenous | fully human | EGFR | Metastatic squamous non-small cell lung carcinoma | 125547 | Link |
| nivolumab | Opdivo | Bristol-Myers Squibb | 3/4/2015 | intravenous | fully human | PD-1 | Metastatic squamous non-small cell lung carcinoma | 125527 | Link |
| nivolumab | Opdivo | Bristol-Myers Squibb | 12/22/2014 | intravenous | fully human | PD-1 | Metastatic melanoma | 125554 | Link |
| obiltoxaximab | Anthem | Elusys Therapeutics | 3/18/2016 | intravenous | chimeric | Protective antigen of the Anthrax toxin | Inhalational anthrax | 125509 | Link |
| obinutuzumab | Gazyva | Genentech | 11/1/2013 | intravenous | humanized | CD20 | Chronic lymphocytic leukemia | 125486 | Link |
| ocrelizumab | Ocrevus | Genentech | 3/28/2017 | intravenous | humanized | CD20 | Multiple sclerosis | 761053 | Link |
| ofatumumab | Arzerra | Glaxo Grp | 10/26/2009 | intravenous | fully human | CD20 | Chronic lymphocytic leukemia | 125326 | Link |
| olaratumab | Lartruvo | Eli Lilly | 10/19/2016 | intravenous | fully human | PDGFRA | Soft tissue sarcoma | 761038 | Link |
| omalizumab | Xolair | Genentech | 6/20/2003 | subcutaneous | humanized | IgE | Moderate to severe persistent asthma | 103976 | Link |
| palivizumab | Synagis | MedImmune | 6/19/1998 | intramuscular | humanized | F protein of RSV | Respiratory syncytial virus | 103770 | Link |
| panitumumab | Vectibix | Amgen | 9/27/2006 | intravenous | fully human | EGFR | Metastatic colorectal cancer | 125147 | Link |
| pembrolizumab | Keytruda | Merck | 9/4/2014 | intravenous | humanized | PD-1 | Metastatic melanoma | 125514 | Link |
| pertuzumab | Perjeta | Genentech | 6/8/2012 | intravenous | humanized | HER2 | Metastatic breast cancer | 125409 | Link |
| ramucirumab | Cyramza | Eli Lilly | 4/21/2014 | intravenous | fully human | VEGFR2 | Gastric cancer | 125477 | Link |
| ranibizumab | Lucentis | Genentech | 6/30/2006 | intravitreal injection | humanized | VEGFR1 VEGFR2 |
Wet age-related macular degeneration | 125156 | Link |
| raxibacumab | Raxibacumab | Human Genome Sciences | 12/24/2012 | intravenous | fully human | Protective antigen of Bacillus anthracis | Inhalational anthrax | 125349 | Link |
| reslizumab | Cinqair | Teva | 3/23/2016 | intravenous | humanized | IL5 | Severe asthma | 761033 | Link |
| rituximab | Rituxan | Genentech | 11/26/1997 | intravenous | chimeric | CD20 | B-cell non-Hodgkin's lymphoma | 103705 | Link |
| rituximab and hyaluronidase | Rituxan Hycela | Genentech | 6/22/17 | subcutaneous | chimeric, co-formulated | CD20 | Follicular lymphoma Diffuse large B-cell lymphoma Chronic lymphocytic leukemia |
761064 | Link |
| sarilumab | Kevzara | Sanofi Aventis | 5/22/17 | subcutaneous | fully human | IL6R | Rheumatoid arthritis | 761037 | Link |
| secukinumab | Cosentyx | Novartis | 1/21/2015 | subcutaneous (2015) intravenous (2023) |
fully human | IL17A | Plaque psoriasis Ankylosing spondylitis |
125504 | Link |
| siltuximab | Sylvant | Janssen Biotech | 4/23/2014 | intravenous | chimeric | IL6 | Multicentric Castleman's disease | 125496 | Link |
| tildrakizumab-asmn | Ilumya | Merck | 3/20/18 | subcutaneous | humanized | IL23 | Plaque psoriasis | 761067 | Link |
| tocilizumab | Actemra | Genentech | 1/8/2010 | intravenous | humanized | IL6R | Rheumatoid arthritis | 125276 | Link |
| tocilizumab | Actemra | Genentech | 10/21/2013 | intravenous subcutaneous |
humanized | IL6R | Rheumatoid arthritis Polyarticular juvenile idiopathic arthritis Systemic juvenile idiopathic arthritis |
125472 | Link |
| trastuzumab | Herceptin | Genentech | 9/25/1998 | intravenous | humanized | HER2 | Metastatic breast cancer | 103792 | Link |
| trastuzumab-dkst | Ogivri | Mylan | 12/1/17 | intravenous | humanized, biosimilar | HER2 | HER2-overexpressing breast cancer, metaststic gastric or gastroesophageal junction adenocarcinoma | 761074 | Link |
| ustekinumab | Stelara | Centocor | 9/25/2009 | subcutaneous | fully human | IL12 IL23 |
Plaque psoriasis | 125261 | Link |
| ustekinumab | Stelara | Janssen Biotech | 9/23/2016 | subcutaneous intravenous |
fully human | IL12 IL23 |
Plaque psoriasis Psoriatic arthritis Crohn's disease |
761044 | Link |
| vedolizumab | Entyvio | Takeda | 5/20/2014 | intravenous | humanized | integrin receptor | Ulcerative colitis Crohn's disease |
125476 | Link |
Tositumomab – Bexxar – 2003 – CD20
Mogamulizumab – Poteligeo – August 2018 – CCR4
Moxetumomab pasudotox – Lumoxiti – September 2018 – CD22
Cemiplimab – Libtayo – September 2018 – PD-1
Polatuzumab vedotin – Polivy – June 2019 – CD79B
The bispecific antibodies have arrived in the clinic. In 2009, the bispecific antibody catumaxomab was approved in the European Union[40][41] and was later withdrawn for commercial reasons.[42] Others include amivantamab, blinatumomab, teclistamab, and emicizumab.[43]
Economics
editSince 2000, the therapeutic market for monoclonal antibodies has grown exponentially. In 2006, the "big 5" therapeutic antibodies on the market were bevacizumab, trastuzumab (both oncology), adalimumab, infliximab (both autoimmune and inflammatory disorders, 'AIID') and rituximab (oncology and AIID) accounted for 80% of revenues in 2006. In 2007, eight of the 20 best-selling biotechnology drugs in the U.S. are therapeutic monoclonal antibodies.[44] This rapid growth in demand for monoclonal antibody production has been well accommodated by the industrialization of mAb manufacturing.[45]
References
edit- ^ a b Yao S, Zhu Y, Chen L (February 2013). "Advances in targeting cell surface signalling molecules for immune modulation". Nature Reviews. Drug Discovery. 12 (2): 130–146. doi:10.1038/nrd3877. PMC 3698571. PMID 23370250.
- ^ Janeway, Charles; Paul Travers; Mark Walport; Mark Shlomchik (2001). Immunobiology; Fifth Edition. New York and London: Garland Science. ISBN 978-0-8153-4101-7.
- ^ a b Janeway CA Jr.; et al. (2005). Immunobiology (6th ed.). Garland Science. ISBN 978-0-443-07310-6.
- ^ a b Baxter D (December 2007). "Active and passive immunity, vaccine types, excipients and licensing". Occupational Medicine. 57 (8): 552–556. doi:10.1093/occmed/kqm110. PMID 18045976.
- ^ Modified from Carter P (November 2001). "Improving the efficacy of antibody-based cancer therapies". Nature Reviews. Cancer. 1 (2): 118–129. doi:10.1038/35101072. PMID 11905803. S2CID 10169378.
- ^ Breedveld FC (February 2000). "Therapeutic monoclonal antibodies". Lancet. 355 (9205): 735–740. doi:10.1016/S0140-6736(00)01034-5. PMID 10703815. S2CID 43781004.
- ^ Köhler G, Milstein C (August 1975). "Continuous cultures of fused cells secreting antibody of predefined specificity". Nature. 256 (5517): 495–497. Bibcode:1975Natur.256..495K. doi:10.1038/256495a0. PMID 1172191. S2CID 4161444.
- ^ Nadler LM, Stashenko P, Hardy R, Kaplan WD, Button LN, Kufe DW, et al. (September 1980). "Serotherapy of a patient with a monoclonal antibody directed against a human lymphoma-associated antigen". Cancer Research. 40 (9): 3147–3154. PMID 7427932.
- ^ Ritz J, Schlossman SF (January 1982). "Utilization of monoclonal antibodies in the treatment of leukemia and lymphoma". Blood. 59 (1): 1–11. doi:10.1182/blood.V59.1.1.1. PMID 7032624.
- ^ a b c Stern M, Herrmann R (April 2005). "Overview of monoclonal antibodies in cancer therapy: present and promise". Critical Reviews in Oncology/Hematology. 54 (1): 11–29. doi:10.1016/j.critrevonc.2004.10.011. PMID 15780905.
- ^ a b c Hudson PJ, Souriau C (January 2003). "Engineered antibodies". Nature Medicine. 9 (1): 129–134. doi:10.1038/nm0103-129. PMID 12514726. S2CID 19243664.
- ^ Carter P, Presta L, Gorman CM, Ridgway JB, Henner D, Wong WL, et al. (May 1992). "Humanization of an anti-p185HER2 antibody for human cancer therapy". Proceedings of the National Academy of Sciences of the United States of America. 89 (10): 4285–4289. Bibcode:1992PNAS...89.4285C. doi:10.1073/pnas.89.10.4285. PMC 49066. PMID 1350088.
- ^ Presta LG, Lahr SJ, Shields RL, Porter JP, Gorman CM, Fendly BM, Jardieu PM (September 1993). "Humanization of an antibody directed against IgE". Journal of Immunology. 151 (5): 2623–2632. doi:10.4049/jimmunol.151.5.2623. PMID 8360482. S2CID 904440.
- ^ Chothia C, Lesk AM, Tramontano A, Levitt M, Smith-Gill SJ, Air G, et al. (1989). "Conformations of immunoglobulin hypervariable regions". Nature. 342 (6252): 877–883. Bibcode:1989Natur.342..877C. doi:10.1038/342877a0. PMID 2687698. S2CID 4241051.
- ^ Waldmann TA (March 2003). "Immunotherapy: past, present and future". Nature Medicine. 9 (3): 269–277. doi:10.1038/nm0303-269. PMID 12612576. S2CID 9745527.
- ^ Jefferis R, Lefranc MP (July–August 2009). "Human immunoglobulin allotypes: possible implications for immunogenicity". mAbs. 1 (4): 332–338. doi:10.4161/mabs.1.4.9122. PMC 2726606. PMID 20073133.
- ^ Chapman K, Pullen N, Coney L, Dempster M, Andrews L, Bajramovic J, et al. (2009). "Preclinical development of monoclonal antibodies: considerations for the use of non-human primates". mAbs. 1 (5): 505–516. doi:10.4161/mabs.1.5.9676. PMC 2759500. PMID 20065651.
- ^ Vennepureddy A, Singh P, Rastogi R, Atallah JP, Terjanian T (October 2017). "Evolution of ramucirumab in the treatment of cancer - A review of literature". Journal of Oncology Pharmacy Practice. 23 (7): 525–539. doi:10.1177/1078155216655474. PMID 27306885. S2CID 21298489.
- ^ de Zwart V, Gouw SC, Meyer-Wentrup FA (January 2016). "Antibody therapies for lymphoma in children". The Cochrane Database of Systematic Reviews. 2016 (1): CD011181. doi:10.1002/14651858.cd011181.pub2. PMC 8719646. PMID 26784573.
- ^ a b Sharma P, Allison JP (April 2015). "The future of immune checkpoint therapy". Science. 348 (6230): 56–61. Bibcode:2015Sci...348...56S. doi:10.1126/science.aaa8172. PMID 25838373. S2CID 4608450.
- ^ Dempke WC, Fenchel K, Uciechowski P, Dale SP (March 2017). "Second- and third-generation drugs for immuno-oncology treatment-The more the better?". European Journal of Cancer. 74: 55–72. doi:10.1016/j.ejca.2017.01.001. PMID 28335888.
- ^ a b Rang, H. P. (2003). Pharmacology. Edinburgh: Churchill Livingstone. p. 241. ISBN 978-0-443-07145-4.
- ^ a b Pul R, Dodel R, Stangel M (March 2011). "Antibody-based therapy in Alzheimer's disease". Expert Opinion on Biological Therapy. 11 (3): 343–357. doi:10.1517/14712598.2011.552884. PMID 21261567. S2CID 19375883.
- ^ a b c d van Dyck CH (February 2018). "Anti-Amyloid-β Monoclonal Antibodies for Alzheimer's Disease: Pitfalls and Promise". Biological Psychiatry. 83 (4): 311–319. doi:10.1016/j.biopsych.2017.08.010. PMC 5767539. PMID 28967385.
- ^ a b c d e Guo X, Yan L, Zhang D, Zhao Y (February 2024). "Passive immunotherapy for Alzheimer's disease". Ageing Research Reviews. 94: 102192. doi:10.1016/j.arr.2024.102192. PMID 38219962.
- ^ a b c d Panza F, Solfrizzi V, Imbimbo BP, Logroscino G (October 2014). "Amyloid-directed monoclonal antibodies for the treatment of Alzheimer's disease: the point of no return?". Expert Opinion on Biological Therapy. 14 (10): 1465–1476. doi:10.1517/14712598.2014.935332. PMID 24981190. S2CID 26323381.
- ^ van Dyck CH (February 2018). "Anti-Amyloid-β Monoclonal Antibodies for Alzheimer's Disease: Pitfalls and Promise". Biological Psychiatry. 83 (4): 311–319. doi:10.1016/j.biopsych.2017.08.010. PMC 5767539. PMID 28967385.
- ^ Goel, Ayush (20 August 2013). "Vasogenic cerebral oedema". radiopaedia.org. Retrieved 2017-11-01.
- ^ a b Panza F, Frisardi V, Imbimbo BP, D'Onofrio G, Pietrarossa G, Seripa D, et al. (November 2010). "Bapineuzumab: anti-β-amyloid monoclonal antibodies for the treatment of Alzheimer's disease". Immunotherapy. 2 (6): 767–782. doi:10.2217/imt.10.80. PMID 21091109.
- ^ Sperling RA, Donohue MC, Raman R, Rafii MS, Johnson K, Masters CL, et al. (September 2023). "Trial of Solanezumab in Preclinical Alzheimer's Disease". The New England Journal of Medicine. 389 (12): 1096–1107. doi:10.1056/NEJMoa2305032. PMC 10559996. PMID 37458272.
- ^ Logovinsky V, Satlin A, Lai R, Swanson C, Kaplow J, Osswald G, et al. (April 2016). "Safety and tolerability of BAN2401--a clinical study in Alzheimer's disease with a protofibril selective Aβ antibody". Alzheimer's Research & Therapy. 8 (1): 14. doi:10.1186/s13195-016-0181-2. PMC 4822297. PMID 27048170.
- ^ "A Study to Confirm Safety and Efficacy of BAN2401 in Participants With Early Alzheimer's Disease". Case Medical Research. 2019-03-25. doi:10.31525/ct1-nct03887455. ISSN 2643-4652. S2CID 242999976.
- ^ van Dyck CH, Swanson CJ, Aisen P, Bateman RJ, Chen C, Gee M, et al. (January 2023). "Lecanemab in Early Alzheimer's Disease". The New England Journal of Medicine. 388 (1): 9–21. doi:10.1056/NEJMoa2212948. PMID 36449413.
- ^ "Leqembi | ALZFORUM". www.alzforum.org. Retrieved 2024-02-14.
- ^ Commissioner, Office of the (2023-07-07). "FDA Converts Novel Alzheimer's Disease Treatment to Traditional Approval". FDA. Retrieved 2024-02-14.
- ^ Francis RJ, Sharma SK, Springer C, Green AJ, Hope-Stone LD, Sena L, et al. (September 2002). "A phase I trial of antibody directed enzyme prodrug therapy (ADEPT) in patients with advanced colorectal carcinoma or other CEA producing tumours". British Journal of Cancer. 87 (6): 600–607. doi:10.1038/sj.bjc.6600517. PMC 2364249. PMID 12237768.
- ^ Krauss WC, Park JW, Kirpotin DB, Hong K, Benz CC (2000). "Emerging antibody-based HER2 (ErbB-2/neu) therapeutics". Breast Disease. 11: 113–124. doi:10.3233/bd-1999-11110. PMID 15687597.
- ^ Joyce JA, Fearon DT (April 2015). "T cell exclusion, immune privilege, and the tumor microenvironment". Science. 348 (6230): 74–80. Bibcode:2015Sci...348...74J. doi:10.1126/science.aaa6204. PMID 25838376. S2CID 11603692.
- ^ Hooks MA, Wade CS, Millikan WJ (1991). "Muromonab CD-3: a review of its pharmacology, pharmacokinetics, and clinical use in transplantation". Pharmacotherapy. 11 (1): 26–37. doi:10.1002/j.1875-9114.1991.tb03595.x. PMID 1902291. S2CID 25271222.
- ^ Chames P, Baty D (2009). "Bispecific antibodies for cancer therapy: the light at the end of the tunnel?". mAbs. 1 (6): 539–547. doi:10.4161/mabs.1.6.10015. PMC 2791310. PMID 20073127.
- ^ Linke R, Klein A, Seimetz D (2010). "Catumaxomab: clinical development and future directions". mAbs. 2 (2): 129–136. doi:10.4161/mabs.2.2.11221. PMC 2840231. PMID 20190561.
- ^ "Removab: Withdrawal of the marketing authorisation in the European Union" (PDF). European Medicines Agency. 2017-07-10.
- ^ Toledo-Stuardo K, Ribeiro CH, González-Herrera F, Matthies DJ, Le Roy MS, Dietz-Vargas C, Latorre Y, Campos I, Guerra Y, Tello S, Vásquez-Sáez V, Novoa P, Fehring N, González M, Rodríguez-Siza J, Vásquez G, Méndez P, Altamirano C, Molina MC (October 2024). "Therapeutic antibodies in oncology: an immunopharmacological overview". Cancer Immunol Immunother. 73 (12): 242. doi:10.1007/s00262-024-03814-2. PMC 11448508. PMID 39358613.
- ^ Scolnik PA (2009). "mAbs: a business perspective". mAbs. 1 (2): 179–184. doi:10.4161/mabs.1.2.7736. PMC 2725420. PMID 20061824.
- ^ Kelley B (2009). "Industrialization of mAb production technology: the bioprocessing industry at a crossroads". mAbs. 1 (5): 443–452. doi:10.4161/mabs.1.5.9448. PMC 2759494. PMID 20065641.