This article needs additional citations for verification. (March 2020) |
Aminophenazone (or aminopyrine, amidopyrine, Pyramidon) is a non-narcotic analgesic substance. It is a pyrazolone and a derivative of phenazone, which also has anti-inflammatory and antipyretic properties. While inexpensive and effective, especially in the treatment of rheumatism, the drug carries a serious risk of severe and sometimes fatal side-effects, including agranulocytosis.[citation needed] While its production and use have been banned in many countries, including France, Thailand, India and Japan, it is still sometimes used in the developing world.[citation needed]
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Pharmacokinetic data | |
| Metabolism | N-demethylation[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.332 |
| Chemical and physical data | |
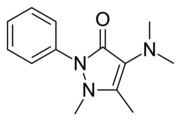
| Formula | C13H17N3O |
| Molar mass | 231.299 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
A breath test with 13C-labeled aminopyrine has been used as a non-invasive measure of cytochrome P-450 metabolic activity in liver-function tests.[1] It is also used in measuring the total body water in the human body system.[2]
History
editAminophenazone was first synthesized by Friedrich Stolz and Ludwig Knorr in the late nineteenth century, and sold as an anti-fever medicine known as Pyramidon by Hoechst AG from 1897 until its eventual replacement[when?] by the safer propyphenazone molecule.
Symptoms
editSymptoms of exposure to this compound include:[3]
- allergic reactions
- strong spasmolytic effect on smooth muscle of peripheral blood vessels
- irritability
- palsy
- copious sweating
- dilated pupils
- sharp drop then rise in body temperature
- dysuria
- dyspnea
- anxiety
- rectal tenesmus
- urinary frequency
- intermittent fever
- fatty infiltration of the liver
- heart muscle degeneration
- death due to circulatory failure following cardiovascular collapse
Agranulocytosis often occurs. Ingestion may cause central nervous system stimulation, vomiting, convulsions, cyanosis, tinnitus, leukopenia, kidney damage and coma. Ingestion may also lead to nausea, mental disturbances, methemoglobinemia, chocolate-colored blood, dizziness, epigastric pain, difficulty in hearing, thready pulse and liver damage.
Other symptoms reported via ingestion include hemolytic anemia, porphyria and severe gastrointestinal bleeding. Bone marrow depression also occurs. Rare eye effects include acute transient myopia.
Chronic symptoms include:
- anorexia
- edema
- oliguria
- urticaria
- hypersensitivity
- aplastic anemia
- sore throat
- fever
- pharyngeal membrane
- jaundice enlargement of the liver and spleen
- exfoliative dermatitis
- gastric or duodenal erosion with perforation or bleeding
- adrenal necrosis
- thrombocytopenic purpura
- acute leukemia
When heated to decomposition this compound emits toxic fumes of nitrogen oxides.
Metabolism
editAmidopyrine is metabolized by demethylation and acetylation. Amidopirina metabolites are 4-aminoantipyrine, metilaminoantipirin, rubazonovaya and metilrubazonovaya acid. These acids have a reddish color. At high amidopirine doses, urine can have a reddish brown coloration, due to the presence in the urine of these acid markers.[4]
In popular culture
editHospital Universitario Ramón y Cajal in Madrid, Spain was built as a center for specialized surgery, a panacea for the ills of the health system in Madrid. Locals hence nicknamed it el piramidón.[5]
References
edit- ^ a b Caubet MS, Laplante A, Caillé J, Brazier JL (June 2002). "[13C]aminopyrine and [13C]caffeine breath test: influence of gender, cigarette smoking and oral contraceptives intake". Isotopes in Environmental and Health Studies. 38 (2): 71–7. Bibcode:2002IEHS...38...71C. doi:10.1080/10256010208033314. PMID 12219983. S2CID 22002940.
- ^ "Aminophenazone — Compound Summary". PubChem. The National Library of Medicine. 2005-03-26. Retrieved June 12, 2008.
- ^ Pubchem. "aminophenazone". pubchem.ncbi.nlm.nih.gov. Retrieved 2017-11-09.
- ^ Vyzanie. "Amidopirina". soundlike.ru. Archived from the original on 2022-02-01. Retrieved 2017-11-09.
- ^ Bellver C (27 January 1980). "La ubicacion de los hospitales, un ejemplo perfecto de mala planificación sanitaria". El País (in European Spanish).
De acuerdo con todo ello, el Ramón y Cajal comenzó a ser esperado como la gran panacea que tenía que resolver gran parte de los problemas hospitalarios madrileños. Por ello, alguien pensó que, siguiendo con los términos médicos, el citado centro venía a ser a la sanidad madrileña como el Piramidón -un antipirético muy utilizado para eliminar los síntomas de la enfermedad, aunque no la enfermedad en sí- a cualquier enfermo. De ahí a que el Centro Ramón y Cajal pasara ser conocido por todos como el Piramidón no medió ni un paso.