Nitrogen mustards (NMs) are cytotoxic organic compounds with the bis(2-chloroethyl)amino ((ClC2H4)2NR) functional group.[1] Although originally produced as chemical warfare agents,[2][3] they were the first chemotherapeutic agents for treatment of cancer.[4] Nitrogen mustards are nonspecific DNA alkylating agents.
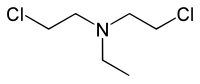


Name
editNitrogen mustards are not related to the mustard plant or its pungent essence, allyl isothiocyanate; the name comes from the pungent smell of chemical weapons preparations.[5]
Chemical warfare
editDuring World War II, nitrogen mustards were studied at the Yale School of Medicine by Alfred Gilman and Louis Goodman, and in December 1942, they started classified human clinical trials of nitrogen mustards for the treatment of lymphoma.[6] In early December of 1943, an incident during the air raid on Bari, Italy, led to the release of mustard gas that affected several hundred soldiers and civilians.[7] Medical examination of the survivors showed a decreased number of lymphocytes.[8] After World War II was over, the Bari incident and the Yale group's studies eventually converged prompting a search for other similar compounds. Due to its use in previous studies, the nitrogen mustard known as "HN2" became the first chemotherapy drug mustine.
Examples
editThe nitrogen mustard drug mustine (HN2), is no longer commonly in use in its original IV formulation because of excessive toxicity. Other nitrogen mustards developed include cyclophosphamide, chlorambucil, uramustine, melphalan, and bendamustine.[9] Bendamustine has recently re-emerged as a viable chemotherapeutic treatment.[10]
Nitrogen mustards that can be used for chemical warfare purposes are tightly regulated. Their weapon designations are:[11]
Normustard (mustine without a methyl group on the nitrogen atom; bis(2-chloroethyl)ethylamine) can be used in the synthesis of piperazine drugs such as mazapertine, aripiprazole & fluanisone. Canfosfamide was also made from normustard.
Some nitrogen mustards of opiates were also prepared, although these are not known to be antineoplastic. Examples include chlornaltrexamine and chloroxymorphamine.
Mechanism of action
editNitrogen mustards form cyclic ammonium ions (aziridinium ions) by intramolecular displacement of the chloride by the amine nitrogen. This aziridinium group then alkylates DNA once it is attacked by the N-7 nucleophilic center on the guanine base. A second attack after the displacement of the second chlorine forms the second alkylation step that results in the formation of interstrand cross-links (ICLs) as it was shown in the early 1960s. At that time, it was proposed that the ICLs were formed between N-7 atom of guanine residue in a 5’-d(GC) sequence.[12][13] Later it was clearly demonstrated that nitrogen mustards form a 1,3 ICL in the 5’-d(GNC) sequence.[14][15][16][17]
The strong cytotoxic effect caused by the formation of ICLs is what makes NMs an effective chemotherapeutic agent. Other compounds used in cancer chemotherapy that have the ability to form ICLs are cisplatin, mitomycin C, carmustine, and psoralen.[18] These kinds of lesions are effective at forcing the cell to undergo apoptosis via p53,[citation needed] a protein which scans the genome for defects. Note that the alkylating damage itself is not cytotoxic and does not directly cause cell death.
Safety
editNitrogen mustards are powerful and persistent blister agents. HN1, HN2, HN3 are therefore classified as Schedule 1 substances within the Chemical Weapons Convention.[19] Production and use is therefore strongly restricted.[20]
See also
editReferences
edit- ^ "Mustards". The IUPAC Compendium of Chemical Terminology. 2014. doi:10.1351/goldbook.M04071.
- ^ Nitrogen mustard gas was stockpiled by several nations during the Second World War, but it was never used in combat.Daniel C. Keyes; Jonathan L. Burstein; Richard B. Schwartz; Raymond E. Swienton (2004). Medical Response to Terrorism: Preparedness and Clinical Practice. Lippincott Williams & Wilkins. p. 16. ISBN 978-0781749862 – via books.google.com.
- ^ Centers for Disease Control and Prevention (April 4, 2013). "Facts About Nitrogen Mustards". cdc.gov. Archived from the original on September 3, 2013. Retrieved September 12, 2013.
- ^ Chabner, Bruce A.; Roberts, Thomas G. (2005). "Chemotherapy and the war on cancer". Nature Reviews Cancer. 5 (1): 65–72. doi:10.1038/nrc1529. PMID 15630416. S2CID 205467419.
- ^ Ghorani-Azam, Adel; Balali-Mood, Mahdi (December 1, 2015). "Clinical Pharmacology and Toxicology of Mustard Compounds". Basic and Clinical Toxicology of Mustard Compounds. Springer Science+Business Media. p. 64. ISBN 9783319238746. Retrieved March 12, 2019 – via Google Books.
- ^ Gilman A (May 1963). "The initial clinical trial of nitrogen mustard". Am. J. Surg. 105 (5): 574–8. doi:10.1016/0002-9610(63)90232-0. PMID 13947966.
- ^ Jules Hirsch, MD; Journal of the American Medical Association (2006). "An Anniversary for Cancer Chemotherapy". JAMA. 296 (12). jamanetwork.com: 1518–1520. doi:10.1001/jama.296.12.1518. PMID 17003400.
- ^ Hirsch J (September 2006). "An anniversary for cancer chemotherapy". JAMA. 296 (12): 1518–20. doi:10.1001/jama.296.12.1518. PMID 17003400.
- ^ Mattes, W. B.; Hartley, J. A.; Kohn, K. W. (1986). "DNA sequence selectivity of guanine–N7 alkylation by nitrogen mustards". Nucleic Acids Research. 14 (7): 2971–2987. doi:10.1093/nar/14.7.2971. PMC 339715. PMID 3960738.
- ^ Cheson BD, Rummel MJ (March 2009). "Bendamustine: rebirth of an old drug". J. Clin. Oncol. 27 (9): 1492–501. doi:10.1200/JCO.2008.18.7252. PMID 19224851.[permanent dead link]
- ^ University of Durham. "SCHEDULE 1 CHEMICALS" (PDF). dur.ac.uk.
- ^ Geiduschek EP (July 1961). ""Reversible" DNA". Proc. Natl. Acad. Sci. U.S.A. 47 (7): 950–5. Bibcode:1961PNAS...47..950G. doi:10.1073/pnas.47.7.950. PMC 221307. PMID 13704192.
- ^ Brookes P, Lawley PD (September 1961). "The reaction of mono- and di-functional alkylating agents with nucleic acids". Biochem. J. 80 (3): 496–503. doi:10.1042/bj0800496. PMC 1243259. PMID 16748923.
- ^ Millard JT, Raucher S, Hopkins PB (1990). "Mechlorethamine Cross Links Deoxyguanosine Residues at 5'-GNC Sequences in Duplex DNA Fragments". Journal of the American Chemical Society. 112 (6): 2459–60. doi:10.1021/ja00162a079.
- ^ Rink SM, Solomon MS, Taylor MJ, Rajur SB, McLaughlin LW, Hopkins PB (1993). "Covalent structure of a nitrogen mustard-induced DNA interstrand cross-link: An N7-to-N7 linkage of deoxyguanosine residues at the duplex sequence 5'-d(GNC)". Journal of the American Chemical Society. 115 (7): 2551–7. doi:10.1021/ja00060a001.
- ^ Dong Q, Barsky D, Colvin ME, et al. (December 1995). "A structural basis for a phosphoramide mustard-induced DNA interstrand cross-link at 5'-d(GAC)". Proc. Natl. Acad. Sci. U.S.A. 92 (26): 12170–4. Bibcode:1995PNAS...9212170D. doi:10.1073/pnas.92.26.12170. PMC 40318. PMID 8618865.
- ^ Bauer GB, Povirk LF (March 1997). "Specificity and kinetics of interstrand and intrastrand bifunctional alkylation by nitrogen mustards at a G-G-C sequence". Nucleic Acids Res. 25 (6): 1211–8. doi:10.1093/nar/25.6.1211. PMC 146567. PMID 9092631.
- ^ Guainazzi, A.; Schärer, O. D. (2010). "Using synthetic DNA interstrand crosslinks to elucidate repair pathways and identify new therapeutic targets for cancer chemotherapy". Cellular and Molecular Life Sciences. 67 (21): 3683–3697. doi:10.1007/s00018-010-0492-6. PMC 3732395. PMID 20730555.
- ^ Organisation for the Prohibition of Chemical Weapons. "Chemical Weapons Convention: Schedule 1 Toxic chemicals". opcw.org. Archived from the original on 2013-06-07.
- ^ United States Department of State, Bureau of Arms Control, Verification and Compliance; United States Department of Commerce, Bureau of Industry and Security (May 2004). "Introduction to Industry Implementation of the Chemical Weapons Convention" (PDF). cwc.gov. Archived from the original (PDF) on 2013-10-20. Retrieved 2013-09-12.
{{cite web}}: CS1 maint: multiple names: authors list (link)
Further reading
edit- Stanford University School of Medicine (2013). "Topical Nitrogen Mustard (Mustargen)". stanford.edu.
- University of California, Los Angeles (2002). "Brassica alba or Sinapis nigra". ucla.edu.