C4 carbon fixation or the Hatch–Slack pathway is one of three known photosynthetic processes of carbon fixation in plants. It owes the names to the 1960s discovery by Marshall Davidson Hatch and Charles Roger Slack.[1]
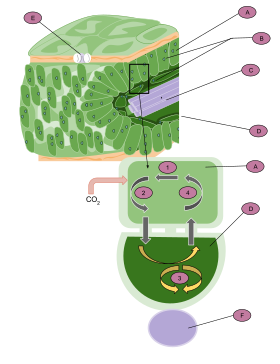
A: Mesophyll cell
B: Chloroplast
C: Vascular tissue
D: Bundle sheath cell
E: Stoma
F: Vascular tissue
1. CO2 is fixed to produce a four-carbon molecule (malate or aspartate).
2. The molecule exits the cell and enters the bundle sheath cells.
3. It is then broken down into CO2 and pyruvate. CO2 enters the Calvin cycle to produce carbohydrates.
4. Pyruvate reenters the mesophyll cell, where it is reused to produce malate or aspartate.
C4 fixation is an addition to the ancestral and more common C3 carbon fixation. The main carboxylating enzyme in C3 photosynthesis is called RuBisCO, which catalyses two distinct reactions using either CO2 (carboxylation) or oxygen (oxygenation) as a substrate. RuBisCO oxygenation gives rise to phosphoglycolate, which is toxic and requires the expenditure of energy to recycle through photorespiration. C4 photosynthesis reduces photorespiration by concentrating CO2 around RuBisCO.
To enable RuBisCO to work in an environment where there is a lot of carbon dioxide and very little oxygen, C4 leaves generally contain two partially isolated compartments called mesophyll cells and bundle-sheath cells. CO2 is initially fixed in the mesophyll cells in a reaction catalysed by the enzyme PEP carboxylase in which the three-carbon phosphoenolpyruvate (PEP) reacts with CO2 to form the four-carbon oxaloacetic acid (OAA). OAA can then be reduced to malate or transaminated to aspartate. These intermediates diffuse to the bundle sheath cells, where they are decarboxylated, creating a CO2-rich environment around RuBisCO and thereby suppressing photorespiration. The resulting pyruvate (PYR), together with about half of the phosphoglycerate (PGA) produced by RuBisCO, diffuses back to the mesophyll. PGA is then chemically reduced and diffuses back to the bundle sheath to complete the reductive pentose phosphate cycle (RPP). This exchange of metabolites is essential for C4 photosynthesis to work.
Additional biochemical steps require more energy in the form of ATP to regenerate PEP, but concentrating CO2 allows high rates of photosynthesis at higher temperatures. Higher CO2 concentration overcomes the reduction of gas solubility with temperature (Henry's law). The CO2 concentrating mechanism also maintains high gradients of CO2 concentration across the stomatal pores. This means that C4 plants have generally lower stomatal conductance, reduced water losses and have generally higher water-use efficiency.[2] C4 plants are also more efficient in using nitrogen, since PEP carboxylase is cheaper to make than RuBisCO.[3] However, since the C3 pathway does not require extra energy for the regeneration of PEP, it is more efficient in conditions where photorespiration is limited, typically at low temperatures and in the shade.[4]
Discovery
editThe first experiments indicating that some plants do not use C3 carbon fixation but instead produce malate and aspartate in the first step of carbon fixation were done in the 1950s and early 1960s by Hugo Peter Kortschak and Yuri Karpilov.[5][6] The C4 pathway was elucidated by Marshall Davidson Hatch and Charles Roger Slack, in Australia, in 1966.[1] While Hatch and Slack originally referred to the pathway as the "C4 dicarboxylic acid pathway", it is sometimes called the Hatch–Slack pathway.[6]
Anatomy
editC4 plants often possess a characteristic leaf anatomy called kranz anatomy, from the German word for wreath. Their vascular bundles are surrounded by two rings of cells; the inner ring, called bundle sheath cells, contains starch-rich chloroplasts lacking grana, which differ from those in mesophyll cells present as the outer ring. Hence, the chloroplasts are called dimorphic. The primary function of kranz anatomy is to provide a site in which CO2 can be concentrated around RuBisCO, thereby avoiding photorespiration. Mesophyll and bundle sheath cells are connected through numerous cytoplasmic sleeves called plasmodesmata whose permeability at leaf level is called bundle sheath conductance. A layer of suberin[7] is often deposed at the level of the middle lamella (tangential interface between mesophyll and bundle sheath) in order to reduce the apoplastic diffusion of CO2 (called leakage). The carbon concentration mechanism in C4 plants distinguishes their isotopic signature from other photosynthetic organisms.
Although most C4 plants exhibit kranz anatomy, there are, however, a few species that operate a limited C4 cycle without any distinct bundle sheath tissue. Suaeda aralocaspica, Bienertia cycloptera, Bienertia sinuspersici and Bienertia kavirense (all chenopods) are terrestrial plants that inhabit dry, salty depressions in the deserts of the Middle East. These plants have been shown to operate single-cell C4 CO2-concentrating mechanisms, which are unique among the known C4 mechanisms.[8][9][10][11] Although the cytology of both genera differs slightly, the basic principle is that fluid-filled vacuoles are employed to divide the cell into two separate areas. Carboxylation enzymes in the cytosol are separated from decarboxylase enzymes and RuBisCO in the chloroplasts. A diffusive barrier is between the chloroplasts (which contain RuBisCO) and the cytosol. This enables a bundle-sheath-type area and a mesophyll-type area to be established within a single cell. Although this does allow a limited C4 cycle to operate, it is relatively inefficient. Much leakage of CO2 from around RuBisCO occurs.
There is also evidence of inducible C4 photosynthesis by non-kranz aquatic macrophyte Hydrilla verticillata under warm conditions, although the mechanism by which CO2 leakage from around RuBisCO is minimised is currently uncertain.[12]
Biochemistry
editIn C3 plants, the first step in the light-independent reactions of photosynthesis is the fixation of CO2 by the enzyme RuBisCO to form 3-phosphoglycerate. However, RuBisCo has a dual carboxylase and oxygenase activity. Oxygenation results in part of the substrate being oxidized rather than carboxylated, resulting in loss of substrate and consumption of energy, in what is known as photorespiration. Oxygenation and carboxylation are competitive, meaning that the rate of the reactions depends on the relative concentration of oxygen and CO2.
In order to reduce the rate of photorespiration, C4 plants increase the concentration of CO2 around RuBisCO. To do so two partially isolated compartments differentiate within leaves, the mesophyll and the bundle sheath. Instead of direct fixation by RuBisCO, CO2 is initially incorporated into a four-carbon organic acid (either malate or aspartate) in the mesophyll. The organic acids then diffuse through plasmodesmata into the bundle sheath cells. There, they are decarboxylated creating a CO2-rich environment. The chloroplasts of the bundle sheath cells convert this CO2 into carbohydrates by the conventional C3 pathway.
There is large variability in the biochemical features of C4 assimilation, and it is generally grouped in three subtypes, differentiated by the main enzyme used for decarboxylation ( NADP-malic enzyme, NADP-ME; NAD-malic enzyme, NAD-ME; and PEP carboxykinase, PEPCK). Since PEPCK is often recruited atop NADP-ME or NAD-ME it was proposed to classify the biochemical variability in two subtypes. For instance, maize and sugarcane use a combination of NADP-ME and PEPCK, millet uses preferentially NAD-ME and Megathyrsus maximus, uses preferentially PEPCK.
NADP-ME
editThe first step in the NADP-ME type C4 pathway is the conversion of pyruvate (Pyr) to phosphoenolpyruvate (PEP), by the enzyme Pyruvate phosphate dikinase (PPDK). This reaction requires inorganic phosphate and ATP plus pyruvate, producing PEP, AMP, and inorganic pyrophosphate (PPi). The next step is the carboxylation of PEP by the PEP carboxylase enzyme (PEPC) producing oxaloacetate. Both of these steps occur in the mesophyll cells:
- pyruvate + Pi + ATP → PEP + AMP + PPi
- PEP + CO2 → oxaloacetate
PEPC has a low KM for HCO−
3 — and, hence, high affinity, and is not confounded by O2 thus it will work even at low concentrations of CO2.
The product is usually converted to malate (M), which diffuses to the bundle-sheath cells surrounding a nearby vein. Here, it is decarboxylated by the NADP-malic enzyme (NADP-ME) to produce CO2 and pyruvate. The CO2 is fixed by RuBisCo to produce phosphoglycerate (PGA) while the pyruvate is transported back to the mesophyll cell, together with about half of the phosphoglycerate (PGA). This PGA is chemically reduced in the mesophyll and diffuses back to the bundle sheath where it enters the conversion phase of the Calvin cycle. For each CO2 molecule exported to the bundle sheath the malate shuttle transfers two electrons, and therefore reduces the demand of reducing power in the bundle sheath.
NAD-ME
editHere, the OAA produced by PEPC is transaminated by aspartate aminotransferase to aspartate (ASP) which is the metabolite diffusing to the bundle sheath. In the bundle sheath ASP is transaminated again to OAA and then undergoes a futile reduction and oxidative decarboxylation to release CO2. The resulting Pyruvate is transaminated to alanine, diffusing to the mesophyll. Alanine is finally transaminated to pyruvate (PYR) which can be regenerated to PEP by PPDK in the mesophyll chloroplasts. This cycle bypasses the reaction of malate dehydrogenase in the mesophyll and therefore does not transfer reducing equivalents to the bundle sheath.
PEPCK
editIn this variant the OAA produced by aspartate aminotransferase in the bundle sheath is decarboxylated to PEP by PEPCK. The fate of PEP is still debated. The simplest explanation is that PEP would diffuse back to the mesophyll to serve as a substrate for PEPC. Because PEPCK uses only one ATP molecule, the regeneration of PEP through PEPCK would theoretically increase photosynthetic efficiency of this subtype, however this has never been measured. An increase in relative expression of PEPCK has been observed under low light, and it has been proposed to play a role in facilitating balancing energy requirements between mesophyll and bundle sheath.
Metabolite exchange
editWhile in C3 photosynthesis each chloroplast is capable of completing light reactions and dark reactions, C4 chloroplasts differentiate in two populations, contained in the mesophyll and bundle sheath cells. The division of the photosynthetic work between two types of chloroplasts results inevitably in a prolific exchange of intermediates between them. The fluxes are large and can be up to ten times the rate of gross assimilation.[13] The type of metabolite exchanged and the overall rate will depend on the subtype. To reduce product inhibition of photosynthetic enzymes (for instance PECP) concentration gradients need to be as low as possible. This requires increasing the conductance of metabolites between mesophyll and bundle sheath, but this would also increase the retro-diffusion of CO2 out of the bundle sheath, resulting in an inherent and inevitable trade off in the optimisation of the CO2 concentrating mechanism.
Light harvesting and light reactions
editTo meet the NADPH and ATP demands in the mesophyll and bundle sheath, light needs to be harvested and shared between two distinct electron transfer chains. ATP may be produced in the bundle sheath mainly through cyclic electron flow around Photosystem I, or in the M mainly through linear electron flow depending on the light available in the bundle sheath or in the mesophyll. The relative requirement of ATP and NADPH in each type of cells will depend on the photosynthetic subtype.[13] The apportioning of excitation energy between the two cell types will influence the availability of ATP and NADPH in the mesophyll and bundle sheath. For instance, green light is not strongly adsorbed by mesophyll cells and can preferentially excite bundle sheath cells, or vice versa for blue light.[14] Because bundle sheaths are surrounded by mesophyll, light harvesting in the mesophyll will reduce the light available to reach BS cells. Also, the bundle sheath size limits the amount of light that can be harvested.[15]
Efficiency
editDifferent formulations of efficiency are possible depending on which outputs and inputs are considered. For instance, average quantum efficiency is the ratio between gross assimilation and either absorbed or incident light intensity. Large variability of measured quantum efficiency is reported in the literature between plants grown in different conditions and classified in different subtypes but the underpinnings are still unclear. One of the components of quantum efficiency is the efficiency of dark reactions, biochemical efficiency, which is generally expressed in reciprocal terms as ATP cost of gross assimilation (ATP/GA).
In C3 photosynthesis ATP/GA depends mainly on CO2 and O2 concentration at the carboxylating sites of RuBisCO. When CO2 concentration is high and O2 concentration is low photorespiration is suppressed and C3 assimilation is fast and efficient, with ATP/GA approaching the theoretical minimum of 3.
In C4 photosynthesis CO2 concentration at the RuBisCO carboxylating sites is mainly the result of the operation of the CO2 concentrating mechanisms, which cost circa an additional 2 ATP/GA but makes efficiency relatively insensitive of external CO2 concentration in a broad range of conditions.
Biochemical efficiency depends mainly on the speed of CO2 delivery to the bundle sheath, and will generally decrease under low light when PEP carboxylation rate decreases, lowering the ratio of CO2/O2 concentration at the carboxylating sites of RuBisCO. The key parameter defining how much efficiency will decrease under low light is bundle sheath conductance. Plants with higher bundle sheath conductance will be facilitated in the exchange of metabolites between the mesophyll and bundle sheath and will be capable of high rates of assimilation under high light. However, they will also have high rates of CO2 retro-diffusion from the bundle sheath (called leakage) which will increase photorespiration and decrease biochemical efficiency under dim light. This represents an inherent and inevitable trade off in the operation of C4 photosynthesis. C4 plants have an outstanding capacity to attune bundle sheath conductance. Interestingly, bundle sheath conductance is downregulated in plants grown under low light[16] and in plants grown under high light subsequently transferred to low light as it occurs in crop canopies where older leaves are shaded by new growth.[17]
Evolution and advantages
editC4 plants have a competitive advantage over plants possessing the more common C3 carbon fixation pathway under conditions of drought, high temperatures, and nitrogen or CO2 limitation. When grown in the same environment, at 30 °C, C3 grasses lose approximately 833 molecules of water per CO2 molecule that is fixed, whereas C4 grasses lose only 277. This increased water use efficiency of C4 grasses means that soil moisture is conserved, allowing them to grow for longer in arid environments.[18]
C4 carbon fixation has evolved in at least 62 independent occasions in 19 different families of plants, making it a prime example of convergent evolution.[19][20] This convergence may have been facilitated by the fact that many potential evolutionary pathways to a C4 phenotype exist, many of which involve initial evolutionary steps not directly related to photosynthesis.[21] C4 plants arose around 35 million years ago[20] during the Oligocene (precisely when is difficult to determine) and were becoming ecologically significant in the early Miocene around 21 million years ago.[22] C4 metabolism in grasses originated when their habitat migrated from the shady forest undercanopy to more open environments,[23] where the high sunlight gave it an advantage over the C3 pathway.[24] Drought was not necessary for its innovation; rather, the increased parsimony in water use was a byproduct of the pathway and allowed C4 plants to more readily colonize arid environments.[24]
Today, C4 plants represent about 5% of Earth's plant biomass and 3% of its known plant species.[18][25] Despite this scarcity, they account for about 23% of terrestrial carbon fixation.[26][27] Increasing the proportion of C4 plants on earth could assist biosequestration of CO2 and represent an important climate change avoidance strategy. Present-day C4 plants are concentrated in the tropics and subtropics (below latitudes of 45 degrees) where the high air temperature increases rates of photorespiration in C3 plants.
Plants that use C4 carbon fixation
editAbout 8,100 plant species use C4 carbon fixation, which represents about 3% of all terrestrial species of plants.[27][28] All these 8,100 species are angiosperms. C4 carbon fixation is more common in monocots compared with dicots, with 40% of monocots using the C4 pathway[clarification needed], compared with only 4.5% of dicots. Despite this, only three families of monocots use C4 carbon fixation compared to 15 dicot families. Of the monocot clades containing C4 plants, the grass (Poaceae) species use the C4 photosynthetic pathway most. 46% of grasses are C4 and together account for 61% of C4 species. C4 has arisen independently in the grass family some twenty or more times, in various subfamilies, tribes, and genera,[29] including the Andropogoneae tribe which contains the food crops maize, sugar cane, and sorghum. Various kinds of millet are also C4.[30][31] Of the dicot clades containing C4 species, the order Caryophyllales contains the most species. Of the families in the Caryophyllales, the Chenopodiaceae use C4 carbon fixation the most, with 550 out of 1,400 species using it. About 250 of the 1,000 species of the related Amaranthaceae also use C4.[18][32]
Members of the sedge family Cyperaceae, and members of numerous families of eudicots – including Asteraceae (the daisy family), Brassicaceae (the cabbage family), and Euphorbiaceae (the spurge family) – also use C4.
No large trees (above 15 m in height) use C4,[33] however a number of small trees or shrubs smaller than 10 m exist which do: six species of Euphorbiaceae all native to Hawaii and two species of Amaranthaceae growing in deserts of the Middle-East and Asia.[34]
Converting C3 plants to C4
editGiven the advantages of C4, a group of scientists from institutions around the world are working on the C4 Rice Project to produce a strain of rice, naturally a C3 plant, that uses the C4 pathway by studying the C4 plants maize and Brachypodium.[35] As rice is the world's most important human food—it is the staple food for more than half the planet—having rice that is more efficient at converting sunlight into grain could have significant global benefits towards improving food security. The team claims C4 rice could produce up to 50% more grain—and be able to do it with less water and nutrients.[36][37][38]
The researchers have already identified genes needed for C4 photosynthesis in rice and are now looking towards developing a prototype C4 rice plant. In 2012, the Government of the United Kingdom along with the Bill & Melinda Gates Foundation provided US$14 million over three years towards the C4 Rice Project at the International Rice Research Institute.[39] In 2019, the Bill & Melinda Gates Foundation granted another US$15 million to the Oxford-University-led C4 Rice Project. The goal of the 5-year project is to have experimental field plots up and running in Taiwan by 2024.[40]
C2 photosynthesis, an intermediate step between C3 and Kranz C4, may be preferred over C4 for rice conversion. The simpler system is less optimized for high light and high temperature conditions than C4, but has the advantage of requiring fewer steps of genetic engineering and performing better than C3 under all temperatures and light levels.[41] In 2021, the UK Government provided £1.2 million on studying C2 engineering.[42]
See also
editReferences
edit- ^ a b Slack CR, Hatch MD (June 1967). "Comparative studies on the activity of carboxylases and other enzymes in relation to the new pathway of photosynthetic carbon dioxide fixation in tropical grasses". The Biochemical Journal. 103 (3): 660–5. doi:10.1042/bj1030660. PMC 1270465. PMID 4292834.
- ^ Osborne CP, Sack L (February 2012). "Evolution of C4 plants: a new hypothesis for an interaction of CO2 and water relations mediated by plant hydraulics". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 367 (1588): 583–600. doi:10.1098/rstb.2011.0261. PMC 3248710. PMID 22232769.
- ^ Sage RF, Pearcy RW (July 1987). "The Nitrogen Use Efficiency of C(3) and C(4) Plants: I. Leaf Nitrogen, Growth, and Biomass Partitioning in Chenopodium album (L.) and Amaranthus retroflexus (L.)". Plant Physiology. 84 (3): 954–8. doi:10.1104/pp.84.3.954. PMC 1056701. PMID 16665550.
- ^ Bellasio C, Farquhar GD (July 2019). "3 rice: gains, losses and metabolite fluxes". The New Phytologist. 223 (1): 150–166. doi:10.1111/nph.15787. hdl:1885/159508. PMID 30859576.
- ^ Nickell LG (February 1993). "A tribute to Hugo P. Kortschak: The man, the scientist and the discoverer of C4 photosynthesis". Photosynthesis Research. 35 (2): 201–4. Bibcode:1993PhoRe..35..201N. doi:10.1007/BF00014751. PMID 24318687. S2CID 40107210.
- ^ a b Hatch MD (2002). "C4 photosynthesis: discovery and resolution". Photosynthesis Research. 73 (1–3): 251–6. doi:10.1023/A:1020471718805. PMID 16245128. S2CID 343310.
- ^ Laetsch (1971). Hatch; Osmond; Slatyer (eds.). Photosynthesis and Photorespiration. New York, Wiley-Interscience. ISBN 9780471359005.
- ^ Freitag H, Stichler W (2000). "A remarkable new leaf type with unusual photosynthetic tissue in a central Asiatic genus of Chenopodiaceae". Plant Biology. 2 (2): 154–160. Bibcode:2000PlBio...2..154F. doi:10.1055/s-2000-9462. S2CID 260250537.
- ^ Voznesenskaya EV, Franceschi VR, Kiirats O, Artyusheva EG, Freitag H, Edwards GE (September 2002). "Proof of C4 photosynthesis without Kranz anatomy in Bienertia cycloptera (Chenopodiaceae)". The Plant Journal. 31 (5): 649–62. doi:10.1046/j.1365-313X.2002.01385.x. PMID 12207654. S2CID 14742876.
- ^ Akhani H, Barroca J, Koteeva N, Voznesenskaya E, Franceschi V, Edwards G, Ghaffari SM, Ziegler H (2005). "Bienertia sinuspersici (Chenopodiaceae): A New Species from Southwest Asia and Discovery of a Third Terrestrial C4 Plant Without Kranz Anatomy". Systematic Botany. 30 (2): 290–301. doi:10.1600/0363644054223684. S2CID 85946307.
- ^ Akhani H, Chatrenoor T, Dehghani M, Khoshravesh R, Mahdavi P, Matinzadeh Z (2012). "A new species of Bienertia (Chenopodiaceae) from Iranian salt deserts: a third species of the genus and discovery of a fourth terrestrial C4 plant without Kranz anatomy". Plant Biosystems. 146: 550–559. doi:10.1080/11263504.2012.662921. S2CID 85377740.
- ^ Holaday AS, Bowes G (February 1980). "C(4) Acid Metabolism and Dark CO(2) Fixation in a Submersed Aquatic Macrophyte (Hydrilla verticillata)". Plant Physiology. 65 (2): 331–5. doi:10.1104/pp.65.2.331. PMC 440321. PMID 16661184.
- ^ a b Bellasio C (January 2017). "A generalized stoichiometric model of C3, C2, C2+C4, and C4 photosynthetic metabolism". Journal of Experimental Botany. 68 (2): 269–282. doi:10.1093/jxb/erw303. PMC 5853385. PMID 27535993.
- ^ Evans JR, Vogelmann TC, von Caemmerer S (1 March 2008), "Balancing light capture with distributed metabolic demand during C4 photosynthesis", Charting New Pathways to C4 Rice, WORLD SCIENTIFIC, pp. 127–143, doi:10.1142/9789812709523_0008, ISBN 978-981-270-951-6, retrieved 12 October 2020
- ^ Bellasio C, Lundgren MR (October 2016). "Anatomical constraints to C4 evolution: light harvesting capacity in the bundle sheath". The New Phytologist. 212 (2): 485–96. doi:10.1111/nph.14063. PMID 27375085.
- ^ Bellasio C, Griffiths H (May 2014). "Acclimation to low light by C4 maize: implications for bundle sheath leakiness". Plant, Cell & Environment. 37 (5): 1046–58. doi:10.1111/pce.12194. PMID 24004447.
- ^ Bellasio C, Griffiths H (July 2014). "Acclimation of C4 metabolism to low light in mature maize leaves could limit energetic losses during progressive shading in a crop canopy". Journal of Experimental Botany. 65 (13): 3725–36. doi:10.1093/jxb/eru052. PMC 4085954. PMID 24591058.
- ^ a b c Sage R, Monson R (1999). "7". C4 Plant Biology. Elsevier. pp. 228–229. ISBN 978-0-12-614440-6.
- ^ Sage RF, Christin PA, Edwards EJ (2011). "The C(4) plant lineages of planet Earth". J Exp Bot. 62 (9): 3155–69. doi:10.1093/jxb/err048. PMID 21414957.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Sage RF (1 February 2004). "The evolution of C4 photosynthesis". New Phytologist. 161 (2): 341–370. doi:10.1111/j.1469-8137.2004.00974.x. ISSN 1469-8137. PMID 33873498.
- ^ Williams BP, Johnston IG, Covshoff S, Hibberd JM (September 2013). "Phenotypic landscape inference reveals multiple evolutionary paths to C4 photosynthesis". eLife. 2: e00961. doi:10.7554/eLife.00961. PMC 3786385. PMID 24082995.
- ^ Wooded Grasslands Flourished in Africa 21 Million Years Ago – New Research Forces a Rethink of Ape Evolution
- ^ Edwards EJ, Smith SA (February 2010). "Phylogenetic analyses reveal the shady history of C4 grasses". Proceedings of the National Academy of Sciences of the United States of America. 107 (6): 2532–7. Bibcode:2010PNAS..107.2532E. doi:10.1073/pnas.0909672107. PMC 2823882. PMID 20142480.
- ^ a b Osborne CP, Freckleton RP (May 2009). "Ecological selection pressures for C4 photosynthesis in the grasses". Proceedings. Biological Sciences. 276 (1663): 1753–60. doi:10.1098/rspb.2008.1762. PMC 2674487. PMID 19324795.
- ^ Bond WJ, Woodward FI, Midgley GF (February 2005). "The global distribution of ecosystems in a world without fire". The New Phytologist. 165 (2): 525–37. doi:10.1111/j.1469-8137.2004.01252.x. PMID 15720663. S2CID 4954178.
- ^ Osborne CP, Beerling DJ (January 2006). "Nature's green revolution: the remarkable evolutionary rise of C4 plants". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 361 (1465): 173–94. doi:10.1098/rstb.2005.1737. PMC 1626541. PMID 16553316.
- ^ a b Kellogg EA (July 2013). "C4 photosynthesis". Current Biology. 23 (14): R594-9. doi:10.1016/j.cub.2013.04.066. PMID 23885869.
- ^ Sage RF (July 2016). "A portrait of the C4 photosynthetic family on the 50th anniversary of its discovery: species number, evolutionary lineages, and Hall of Fame". Journal of Experimental Botany. 67 (14): 4039–56. doi:10.1093/jxb/erw156. PMID 27053721.
- ^ Grass Phylogeny Working Group II (January 2012). "New grass phylogeny resolves deep evolutionary relationships and discovers C4 origins". The New Phytologist. 193 (2): 304–12. doi:10.1111/j.1469-8137.2011.03972.x. hdl:2262/73271. PMID 22115274.
- ^ Sage R, Monson R (1999). "16". C4 Plant Biology. Elsevier. pp. 551–580. ISBN 978-0-12-614440-6.
- ^ Zhu XG, Long SP, Ort DR (April 2008). "What is the maximum efficiency with which photosynthesis can convert solar energy into biomass?". Current Opinion in Biotechnology. 19 (2): 153–9. doi:10.1016/j.copbio.2008.02.004. PMID 18374559. Archived from the original on 1 April 2019. Retrieved 29 December 2018.
- ^ Kadereit G, Borsch T, Weising K, Freitag H (2003). "Phylogeny of Amaranthaceae and Chenopodiaceae and the Evolution of C4 Photosynthesis". International Journal of Plant Sciences. 164 (6): 959–986. doi:10.1086/378649. S2CID 83564261.
- ^ Sage, R.F. (May 2001). "Environmental and Evolutionary Preconditions for the Origin and Diversification of the C4 Photosynthetic Syndrome". Plant Biology. 3 (3): 202–213. Bibcode:2001PlBio...3..202S. doi:10.1055/s-2001-15206.
- ^ Young, Sophie N R; Sack, Lawren; Sporck-Koehler, Margaret J; Lundgren, Marjorie R (6 August 2020). "Why is C4 photosynthesis so rare in trees?". Journal of Experimental Botany. 71 (16): 4629–4638. doi:10.1093/jxb/eraa234. PMC 7410182. PMID 32409834.
- ^ Slewinski TL, Anderson AA, Zhang C, Turgeon R (December 2012). "Scarecrow plays a role in establishing Kranz anatomy in maize leaves". Plant & Cell Physiology. 53 (12): 2030–7. doi:10.1093/pcp/pcs147. PMID 23128603.
- ^ Gilles van Kote (24 January 2012). "Researchers aim to flick the high-carbon switch on rice". The Guardian. Retrieved 10 November 2012.
- ^ von Caemmerer S, Quick WP, Furbank RT (June 2012). "The development of C4 rice: current progress and future challenges". Science. 336 (6089): 1671–2. Bibcode:2012Sci...336.1671V. doi:10.1126/science.1220177. PMID 22745421. S2CID 24534351.
- ^ Hibberd JM, Sheehy JE, Langdale JA (April 2008). "Using C4 photosynthesis to increase the yield of rice-rationale and feasibility". Current Opinion in Plant Biology. 11 (2): 228–31. doi:10.1016/j.pbi.2007.11.002. PMID 18203653.
- ^ Hasan M (6 November 2012). "C4 rice project gets financial boost". The News. Archived from the original on 10 November 2012. Retrieved 10 November 2012.
- ^ "Rice to feed the world given a funding boost". University of Oxford. 3 December 2019. Retrieved 29 January 2022.
- ^ Bellasio, Chandra; Farquhar, Graham D. (July 2019). "A leaf-level biochemical model simulating the introduction of C 2 and C 4 photosynthesis in C 3 rice: gains, losses and metabolite fluxes". New Phytologist. 223 (1): 150–166. doi:10.1111/nph.15787. hdl:1885/159508. PMID 30859576. S2CID 75139004.
- ^ "Enhancing crops with C2 photosynthesis". GtR.