Transition metal dithiocarbamate complexes are coordination complexes containing one or more dithiocarbamate ligand, which are typically abbreviated R2dtc−. Many complexes are known. Several homoleptic derivatives have the formula M(R2dtc)n where n = 2 and 3.[1]
Ligand characteristics
editDithiocarbamates are anions. Because of the pi-donor properties of the amino substituent, the two sulfur centers show enhanced basicity. This situation is represented by the zwitterionic resonance structure that depicts a positive charge on N and negative charges on both sulfurs. This N to C pi-bonding results in partial double bond character. Consequently, barriers to rotational about this bond are elevated. Another consequence of their high basicity, dithiocarbamates often stabilize complexes in uncharacteristically high oxidation state (e.g., Fe(IV), Co(IV), Ni(III), Cu(III)).
Dithiocarbamate salts are easily synthesized. Many primary and secondary amines react with carbon disulfide and sodium hydroxide to form dithiocarbamate salts:[2]
- R2NH + CS2 + NaOH → R2NCS2−Na+ + H2O
A wide variety of secondary amines give the corresponding dtc ligand. Popular amines include dimethylamine (Me2NH), diethylamine (Et2NH), and pyrrolidine ((CH2)4NH).
- Related ligands
Dithiocarbamates are classified as derivatives of dithiocarbamic acid. Their properties as ligands resemble the conjugate bases of many related "1,1-dithioacids":
- Xanthates, ROCS2−
- Dithiophosphates, (RO)2PS2−
- Dithiocarboxylates, RCS2−
Synthetic methods
editCommonly, metal dithiocarbamates are prepared by salt metathesis reactions using alkali metal dithiocarbamates:
- NiCl2 + 2 NaS2CNMe2 → Ni(S2CNMe2)2 + 2 NaCl
In some cases, the dithiocarbamate serves as a reductant, followed by its complexation.[3]
A complementary method entails oxidative addition of thiuram disulfides to low-valent metal complexes:
- Mo(CO)6 + 2 [S2CNMe2]2 → Mo(S2CNMe2)4 + 6 CO
Metal amido complexes, such as tetrakis(dimethylamido)titanium, react with carbon disulfide:
- Ti(NMe2)4 + 4 CS2 → Ti(S2CNMe2)4
Homoleptic complexes
edit- Bis complexes
- nickel bis(dimethyldithiocarbamate), palladium bis(dimethyldithiocarbamate), platinum bis(dimethyldithiocarbamate),[4] all square-planar complexes
- copper bis(diethyldithiocarbamate), a square-planar complex[5]
- Tris complexes
- vanadium tris(diethyldithiocarbamate), an octahedral complex[6]
- chromium tris(diethylditiocarbamate), an octahedral complex[7]
- manganese tris(dimthylthtiocarbamate), an octahedral complex[8]
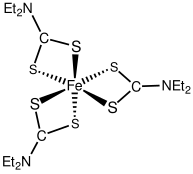
- iron tris(diethyldithiocarbamate), ruthenium tris(diethyldithiocarbamate), osmium tris(diethyldithiocarbamate), all octahedral complexes
- cobalt tris(diethyldithiocarbamate), rhodium tris(diethyldithiocarbamate), iridium tris(diethyldithiocarbamate), all octahedral complexes
- Tetrakis complexes
- titanium tetrakis(dimethyldithiocarbamate)[9]
- molybdenum tetrakis(diethyldithiocarbamate)
- Dimetallic complexes
- iron bis(diethyldithiocarbamate), pentacoordinate Fe dimer
- zinc bis(dimethyldithiocarbamate), pentacoordinate Zn dimer
- dicobalt pentakis(diethyldithiocarbamate) cation, with a pair of octahedral Co(III) centers[10]
- diruthenium pentakis(diethyldithiocarbamate) cation, with a pair of octahedral Ru(III) centers, two isomers
Reactions
editDithiocarbamate complexes do not undergo characteristic reactions. They can be removed from complexes by oxidation, as illustrated by the iodination of the iron tris(diethyldithiocarbamate):
- Fe(S2CNEt2)3 + 0.5 I2 → Fe(S2CNEt2)2I + 0.5 (S2CNEt2)2
They degrade to metal sulfides upon heating.[12]
Applications
editDtc complexes find several applications:
- herbicides in the form of the iron and zinc derivatives Ferbam and Zineb, respectively
- vulcanization accelerators, zinc bis(dimethyldithiocarbamate).[13]
- medicine, iron tris(dimethyldithiocarbamate) as a nitric oxide scavenger.
- lubricants. Metal thiocarbamates are also used in metal-to-metal lubrication proposes, mainly as an anti-oxidation or anti-extreme pressure (EP) additive. 1-2% of such compounds can be added to internal combustion engine lubricant to increase extreme pressure performance in high operational temperatures.[14][15]
References
edit- ^ Coucouvanis, Dimitri (1979). "The Chemistry of the Dithioacid and 1,1-Dithiolate Complexes, 1968–1977". Progress in Inorganic Chemistry. Vol. 26. pp. 301–469. doi:10.1002/9780470166277.ch5. ISBN 9780470166277.
- ^ Rüdiger Schubart (2000). "Dithiocarbamic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a09_001. ISBN 3527306730.
- ^ Ileperuma, O. A.; Feltham, R. D. (1976). "Dithiocarbamate Complexes of Iron and Cobalt Nitrosyls". Inorganic Syntheses. Vol. 16. pp. 5–8. doi:10.1002/9780470132470.ch2. ISBN 978-0-470-13178-7.
- ^ Poirier, Stéphanie; Lynn, Hudson; Reber, Christian; Tailleur, Elodie; Marchivie, Mathieu; Guionneau, Philippe; Probert, Michael R. (2018). "Variation of M···H–C Interactions in Square-Planar Complexes of Nickel(II), Palladium(II), and Platinum(II) Probed by Luminescence Spectroscopy and X-ray Diffraction at Variable Pressure". Inorganic Chemistry. 57 (13): 7713–7723. doi:10.1021/acs.inorgchem.8b00724. PMID 29893549. S2CID 48355413.
- ^ Wang, Chen; Niu, Jiao; Li, Jun; Ma, Xiaoxun (2017). "Synthesis, Structures and Properties of Three Copper Complexes with Dibutyldithiocarbamate Ligand". Journal of Molecular Structure. 1135: 75–81. Bibcode:2017JMoSt1135...75W. doi:10.1016/j.molstruc.2017.01.021.
- ^ Zhu, H.-P.; Deng, Y.-H.; Huang, X.-Y.; Chen, C.-N.; Liu, Q.-T. (1997). "Tris(N,N-diethyldithiocarbamato-S,S')vanadium(III)". Acta Crystallographica Section C Crystal Structure Communications. 53 (6): 692–693. Bibcode:1997AcCrC..53..692Z. doi:10.1107/S0108270196015065.
- ^ Raston, CL; White, AH (1977). "Crystal Structure of Tris(N,N-diethyldithiocarbamato)chromium(III)". Australian Journal of Chemistry. 30 (9): 2091. doi:10.1071/CH9772091.
- ^ Elliot, R. L.; West, B. O.; Snow, M. R.; Tiekink, E. R. T. (1986). "A Second Polymorph of Tris(N,N-diethyldithiocarbamato)manganese(III)". Acta Crystallographica Section C Crystal Structure Communications. 42 (6): 763–764. Bibcode:1986AcCrC..42..763E. doi:10.1107/S0108270186094635.
- ^ Colapietro, M.; Vaciago, A.; Bradley, D. C.; Hursthouse, M. B.; Rendall, I. F. (1972). "Structural studies of metal dithiocarbamates. Part VI. The crystal and molecular structure of tetrakis(NN-diethyldithiocarbamato)titanium(IV)". Journal of the Chemical Society, Dalton Transactions (10): 1052. doi:10.1039/DT9720001052.
- ^ Hendrickson, Alan R.; Martin, Raymond L.; Taylor, Donald (1975). "Synthesis and Properties of Dimeric Cobalt(III) Dithiocarbamate Complexes [Co2(R2dtc)5]+: X-Ray Structural Analysis of Pentakis(diethyldithiocarbamato)dicobalt(III) Tetrafluoroborate". Journal of the Chemical Society, Dalton Transactions (21): 2182. doi:10.1039/dt9750002182.
- ^ Nagy, Eszter M.; Pettenuzzo, Andrea; Boscutti, Giulia; Marchiò, Luciano; Dalla Via, Lisa; Fregona, Dolores (2012). "Ruthenium(II/III)-Based Compounds with Encouraging Antiproliferative Activity against Non-small-Cell Lung Cancer". Chemistry - A European Journal. 18 (45): 14464–14472. doi:10.1002/chem.201202171. PMID 23012112.
- ^ Sarker, Jagodish C.; Hogarth, Graeme (2021). "Dithiocarbamate Complexes as Single Source Precursors to Nanoscale Binary, Ternary and Quaternary Metal Sulfides". Chemical Reviews. 121 (10): 6057–6123. doi:10.1021/acs.chemrev.0c01183. PMID 33847480. S2CID 233222937.
- ^ Engels, Hans-Wilhelm; et al. "Rubber, 4. Chemicals and Additives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a23_365.pub2. ISBN 978-3527306732.
- ^ Shah, Faiz Ullah; Glavatskih, Sergei; Antzutkin, Oleg N. (2012). "Novel Alkylborate–Dithiocarbamate Lubricant Additives: Synthesis and Tribophysical Characterization". Tribology Letters. 45: 67–78. doi:10.1007/s11249-011-9855-x. S2CID 98677629.
- ^ Shekarriz, Marzieh (2016). "An Investigation of Antioxidant Properties of Zinc and Molybdenum Dithiocarbamates in Hydrocarbons". Journal of Petroleum Science and Technology. 6 (2). doi:10.22078/jpst.2016.669.