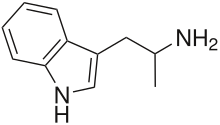
α-Methyltryptamine (αMT, AMT) is a psychedelic, stimulant, and entactogen drug of the tryptamine family.[5][6] It was originally developed as an antidepressant at Upjohn in the 1960s, and was used briefly as an antidepressant in the Soviet Union under the brand name Indopan or Indopane before being discontinued.[4][7][8]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Indopan; Indopane |
| Other names | alpha-Methyltryptamine; αMT; α-ET; AMT; IT-290; IT-403 ((+)-αMT); NSC-97069; PAL-17;[1] Ro 3-0926; U-14,164E; U-14,164-E; 3-(2-Aminopropyl)indole; 3-IT; α-Methyl-3-indoleethanamine; Metryptamine; Amtryptamine |
| Routes of administration | Oral, insufflation, rectal, smoked, IM, IV[2] |
| Drug class | Entactogen; Stimulant; Psychedelic; Hallucinogen; Monoamine releasing agent; Serotonin receptor agonist; Monoamine oxidase inhibitor |
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Onset of action | 3–4 hours[4] |
| Duration of action | 12–24 hours[4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.005.522 |
| Chemical and physical data | |
| Formula | C11H14N2 |
| Molar mass | 174.247 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Side effects of αMT include agitation, restlessness, confusion, lethargy, pupil dilation, jaw clenching, and rapid heart rate, among others.[4][9] αMT acts as a releasing agent of serotonin, norepinephrine, and dopamine, as a serotonin receptor agonist, and as a weak monoamine oxidase inhibitor.[1] αMT is a substituted tryptamine and is closely related to α-ethyltryptamine (αET) and other α-alkylated tryptamines.[1][5]
αMT appears to have first been described by at least 1929.[10][11] It started being more studied in the late 1950s and was briefly used as an antidepressant in the Soviet Union in the 1960s.[4][12][9][13][14] The drug started being used recreationally in the 1960s, with use increasing in the 1990s, and cases of death have been reported.[4][13][9][12] αMT is a controlled substance in various countries, including the United States.[13][4]
Medical uses
editUnder the brand name Indopan or Indopane, αMT at doses of 5 to 10 mg was used for an antidepressant effect.[medical citation needed]
Effects
editWith 20 to 30 mg, euphoria, empathy, and psychedelic effects become apparent and can last as long as 12 hours.[15] A dose exceeding 40 mg is generally considered strong. In rare cases or extreme doses, the duration of effects might exceed 24 hours. Users report that αMT in freebase form is smoked, with doses between and 2 and 5 mg.[2][unreliable source?][5]
Side effects
editNeurologic side effects of αMT include agitation, restlessness, confusion, and lethargy.[4][9] Physical manifestations including vomiting, mydriasis (pupillary dilation), jaw clenching, tachycardia, salivation, diaphoresis (sweating), and elevations in blood pressure, temperature, and respiratory rate.[4][9]
Side effects self-reported by recreational users include anxiety, muscle tension, jaw tightness, pupil dilation, tachycardia, headaches, nausea, and vomiting, as well as psychedelic effects including visual hallucinations and an altered state of mind.[5][16]
αMT is capable of causing life-threatening side effects including hyperthermia, hypertension, and tachycardia.[9][17] Fatalities have been reported in association with high doses or concomitant use of other drugs.[18] Fatalities verified with toxicology and autopsy include those of a 22-year-old man in Miami-Dade county and a British teenager, both of whom died after consuming 1 g of αMT.[19][9]
Pharmacology
editPharmacodynamics
editαMT acts as a relatively balanced reuptake inhibitor and releasing agent of the main three monoamines; serotonin, norepinephrine, and dopamine,[20] and as a non-selective serotonin receptor agonist.[21]
Monoamine oxidase inhibition
editαMT has been shown as a reversible inhibitor of the enzyme monoamine oxidase (MAO) in-vitro[22] and in-vivo.[23]
In rats, the potency of αMT as an MAO-A inhibitor in the brain was approximately equal to that of harmaline at equimolar doses.[note 1] Dextroamphetamine did not enhance the 5-hydroxytryptophan-induced rise of serotonin at any level.[24]
Serotonergic neurotoxicity
editA close analogue of αMT, α-ethyltryptamine (αET), is known to be a serotonergic neurotoxin similarly to MDMA and para-chloroamphetamine (PCA).[25][26][27]
Pharmacokinetics
edit2-Oxo-αMT, 6-hydroxy-αMT, 7-hydroxy-αMT, and 1′-hydroxy-αMT were detected as metabolites of αMT in male Wistar rats.[4][28][29]
Chemistry
editαMT is a synthetic substituted tryptamine with a methyl substituent at the alpha carbon.[1][9] This alpha substitution makes it a relatively poor substrate for monoamine oxidase A, thereby prolonging αMT's half-life, allowing it to reach the brain and enter the central nervous system. Its chemical relation to tryptamine is analogous to that of amphetamine to phenethylamine, amphetamine being α-methylphenethylamine.[9] αMT is closely related to the neurotransmitter serotonin (5-hydroxytryptamine) which partially explains its mechanism of action.
Many analogues of αMT are known, including α-ethyltryptamine (αET), 4-methyl-αMT, 5-chloro-αMT (PAL-542), 5-fluoro-αMT (PAL-544), 5-fluoro-αET (PAL-545), 5-methoxy-αMT (5-MeO-αMT), α,N-dimethyltryptamine (α,N-DMT; N-methyl-αMT), α,N,N-trimethyltryptamine (α,N,N-TMT; N-dimethyl-αMT), α-methylserotonin (α-methyl-5-HT; 5-hydroxy-αMT), and indolylpropylaminopentane (IPAP; α,N-dipropyltryptamine or α,N-DPT), among others.[1][5]
α-Methyltryptophan, a prodrug of α-methylserotonin, also metabolizes into αMT, but only in small amounts.[30][31][32]
Synthesis
editThe synthesis of αMT can be accomplished through several different routes, the two most widely known being the nitroaldol condensation between indole-3-carboxaldehyde and nitroethane under ammonium acetate catalysis which produces 1-(3-indolyl)-2-nitropropene-1, the product can subsequently be reduced using a reducing agent like lithium aluminum hydride[33] It's also hypothesized that it can be reduced using copper(II) chloride and sodium borohydride. The alternative synthesis is the condensation between indole-3-acetone and hydroxylamine.[citation needed], followed by reduction of the obtained ketoxime with lithium aluminum hydride.[5]
History
editαMT has been said to have been first synthesized in 1947, alongside α-ethyltryptamine (αET).[12][26][34] However, other sources suggest that αMT was first described in the scientific literature by at least 1929.[10][11] It was specifically described as an antagonist of ergotamine at this time.[10][11]
αMT started to be more intensively studied, along with αET, in the late 1950s and early 1960s.[12][35][36][37][24][38][39][40][41][42] It was researched by Upjohn (code name U-14,164E) and Sandoz (code name IT-290) as a possible pharmaceutical drug and was simultaneously marketed in the Soviet Union as an antidepressant under the brand name Indopan or Indopane in the 1960s.[13][14][9][43] However, the drug was used clinically for only a short period of time before being withdrawn.[14]
αMT started being used as a recreational drug in the 1960s[9] and use as a designer drug increased in the 1990s.[13] It became a controlled substance in the United States in 2003.[13]
Society and culture
editNames
editαMT never received a formal generic name.[44] In the scientific literature, it has been referred to as α-methyltryptamine or alpha-methyltryptamine (abbreviated as α-MT, αMT, or AMT).[13][14] αMT has also been referred to by developmental code names including IT-290 (Sandoz),[45] NSC-97069,[12] PAL-17,[1] Ro 3-0926,[46][47] and U-14,164E (Upjohn).[48][49][12][9] In the Soviet Union, the drug was merely referred to by its brand name Indopan or Indopane.[50][9] Other synonyms of αMT include 3-(2-aminopropyl)indole and 3-IT.[12] (+)-αMT has been referred to by the code name IT-403.[12][9]
Legality
editAustralia
editThe 5-methoxy analogue, 5-MeO-αMT is schedule 9 in Australia and αMT would be controlled as an analogue of this.[51]
Austria
editαMT is placed under Austrian law (NPSG) Group 6.[52]
Canada
editCanada has no mention of αMT in the Controlled Drugs and Substances Act.[53]
China
editAs of October 2015 αMT is a controlled substance in China.[54]
Denmark
editIn Denmark (2010), the Danish Minister for the Interior and Health placed αMT to their lists of controlled substances (List B).[52]
Finland
editAMT, alfa-methyltryptamine, is a controlled drug in Finland.[55]
Germany
editαMT is listed under the Narcotics Act in schedule 1 (narcotics not eligible for trade and medical prescriptions) in Germany.[52]
Hungary
editαMT was controlled on the Schedule C list in Hungary in 2013.[52]
Lithuania
editIn Lithuania (2012), αMT is controlled as a tryptamine derivative put under control in the 1st list of Narcotic Drugs and Psychotropic Substances which use is prohibited for medical purposes.[52]
Slovakia
editαMT was placed in 2013 on the List of Hazardous Substances in Annex, § 2 in Slovakia.[52]
Slovenia
editαMT appeared on the Decree on Classification of Illicit Drugs in Slovenia (2013).[52]
Spain
editαMT is legal in Spain.[56]
Sweden
editSveriges riksdags health ministry Statens folkhälsoinstitut classified αMT as "health hazard" under the act Lagen om förbud mot vissa hälsofarliga varor (translated Act on the Prohibition of Certain Goods Dangerous to Health) as of Mar 1, 2005, in their regulation SFS 2005:26 listed as alfa-metyltryptamin (AMT), making it illegal to sell or possess.[57]
United Kingdom
editαMT was made illegal in the United Kingdom as of 7 January 2015, along with 5-MeO-DALT.[58] This was following the events of 10 June 2014 when the Advisory Council on the Misuse of Drugs recommended that αMT be scheduled as a class A drug by updating the blanket ban clause on tryptamines.[59]
United States
editThe Drug Enforcement Administration (DEA) placed αMT temporarily in schedule I of the Controlled Substances Act (CSA) on April 4, 2003, pursuant to the temporary scheduling provisions of the CSA (68 FR16427). On September 29, 2004, αMT was permanently controlled as a schedule I substance under the CSA (69FR 58050).[60]
Research
editBesides depression, αMT has been studied in people with schizophrenia and other conditions.[1]
See also
editNotes
editReferences
edit- ^ a b c d e f g Blough BE, Landavazo A, Partilla JS, Decker AM, Page KM, Baumann MH, et al. (October 2014). "Alpha-ethyltryptamines as dual dopamine-serotonin releasers". Bioorganic & Medicinal Chemistry Letters. 24 (19): 4754–4758. doi:10.1016/j.bmcl.2014.07.062. PMC 4211607. PMID 25193229.
- ^ a b Dialtonez (12 March 2003). "AMT FAQ". Erowid Vault.
- ^ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ^ a b c d e f g h i j Barceloux DG (20 March 2012). Medical Toxicology of Drug Abuse: Synthesized Chemicals and Psychoactive Plants. John Wiley & Sons. pp. 196–199. ISBN 978-0-471-72760-6.
- ^ a b c d e f "#48 a-MT". Erowid Online Books: TIHKAL.
- ^ "AMT (alpha-methyltryptamine)". Erowid Vault.
- ^ Iversen L (11 November 2013). Handbook of Psychopharmacology: Volume 14 Affective Disorders: Drug Actions in Animals and Man. Springer Science & Business Media. pp. 132–. ISBN 978-1-4613-4045-4.
- ^ Halberstadt AL, Geyer MA (17 May 2013). "Neuropharmacology of lysergic acid diethylamide (LSD) and other hallucinogens.". In Miller PM, Ball SA, Bates ME, Blume AW, Kampman KM, Kavanagh DJ, Larimer ME, Petry NM, De Witte P (eds.). Biological Research on Addiction: Comprehensive Addictive Behaviors and Disorders. Vol. 2. Academic Press. pp. 625–635 (632). doi:10.1016/B978-0-12-398335-0.00061-3. ISBN 978-0-12-398360-2.
- ^ a b c d e f g h i j k l m n Boland DM, Andollo W, Hime GW, Hearn WL (July–August 2005). "Fatality due to acute alpha-methyltryptamine intoxication". Journal of Analytical Toxicology. 29 (5): 394–397. doi:10.1093/jat/29.5.394. PMID 16105268.
α-Methyltryptamine (AMT) is a synthetic drug of the tryptamine family. It is an indole analogue of amphetamine initially investigated as a monoamine oxidase inhibitor. In the 1960s, the Soviet Union marketed AMT as an antidepressant under the name of Indopan. During the same period, Sandoz (as IT-290 and IT-403) and the Upjohn Company (as [U-14,164E]) studied AMT and its commercial use as a stimulant, but found it to be of little medicinal value. Although clinical use of AMT is obsolete today, recreational use has gained popularity because of the intense hallucinogenic properties lasting up to 16 h. To illustrate recreational use of AMT in the 1960s, Alexander Shulgin, in his book TiHKAL, references the author Ken Kesey and his experiences with AMT and other hallucinogenic drugs (1).
- ^ a b c Gaddum JH, Hameed KA (June 1954). "Drugs which antagonize 5-hydroxytryptamine". British Journal of Pharmacology and Chemotherapy. 9 (2): 240–248. doi:10.1111/j.1476-5381.1954.tb00848.x. PMC 1509436. PMID 13172437.
The antagonism of ergotoxine and tryptamine was described by Laidlaw (1911) and that of ergotamine and α-methyltryptamine by Seki (1929).
- ^ a b c Seki J (1929). "Pharmakologische Untersuchungen des α-Methyl-β-indoläthylamins" [Pharmacological studies of α-methyl-β-indoleethylamine]. Japanese Journal of Medical Sciences. Part 4: Pharmacology. 3. National Research Council of Japan: 235–285. ISSN 0368-3745. OCLC 610325817.
- ^ a b c d e f g h Elliott SP, Brandt SD, Freeman S, Archer RP (March 2013). "AMT (3-(2-aminopropyl)indole) and 5-IT (5-(2-aminopropyl)indole): an analytical challenge and implications for forensic analysis". Drug Testing and Analysis. 5 (3): 196–202. doi:10.1002/dta.1420. PMID 23042766.
α-Methyltryptamine (2, 3-(2-aminopropyl)indole, AMT, α-MT, 3-IT, IT-290, IT-403, U-14, 162-E, Ro 3-0926, NSC 97069, Indopan; Figure 1), on the other hand, is a positional isomer of 5-IT that also shows long-lasting psychoactive effects in humans[1] although further studies are needed to determine the differences or similarities between both psychopharmacological profiles. Following its first synthesis in 1947,[5] the interest in AMT, and other α-alkylated tryptamines, began to develop in the late 1950s when it was discovered that some of these analogues also displayed monoamine oxidase (MAO) inhibiting properties.[6,7] [...]
- ^ a b c d e f g Araújo AM, Carvalho F, Bastos Md, Guedes de Pinho P, Carvalho M (August 2015). "The hallucinogenic world of tryptamines: an updated review". Archives of Toxicology. 89 (8): 1151–1173. doi:10.1007/s00204-015-1513-x. PMID 25877327.
- ^ a b c d Tittarelli R, Mannocchi G, Pantano F, Romolo FS (January 2015). "Recreational use, analysis and toxicity of tryptamines". Current Neuropharmacology. 13 (1): 26–46. doi:10.2174/1570159x13666141210222409. PMC 4462041. PMID 26074742.
- ^ Wilcox J (2012). "Psychoactive properties of alpha-methyltryptamine: analysis from self reports of users". Journal of Psychoactive Drugs. 44 (3): 274–276. doi:10.1080/02791072.2012.704592. PMID 23061328. S2CID 38340985.
- ^ "AMT Effects". Erowid Vault.
- ^ Gillman PK (October 2005). "Monoamine oxidase inhibitors, opioid analgesics and serotonin toxicity". British Journal of Anaesthesia. 95 (4): 434–441. doi:10.1093/bja/aei210. PMID 16051647. "drugs such as MDMA, ecstasy (3,4-methylenedioxymethamphetamine), if combined with MAOIs (including moclobemide) do also cause fatalities because they act as serotonin releasers"
- ^ "Call for ban on drug after reveller's death". Western Gazette. 22 March 2012. Archived from the original on 16 October 2013. Retrieved 1 October 2013.
- ^ "Southampton 'legal high' death deemed 'accidental'". BBC News. 12 November 2013. Retrieved 19 November 2013.
- ^ Nagai F, Nonaka R, Satoh Hisashi Kamimura K (March 2007). "The effects of non-medically used psychoactive drugs on monoamine neurotransmission in rat brain". European Journal of Pharmacology. 559 (2–3): 132–137. doi:10.1016/j.ejphar.2006.11.075. PMID 17223101.
- ^ Nonaka R, Nagai F, Ogata A, Satoh K (December 2007). "In vitro screening of psychoactive drugs by [(35)S]GTPgammaS binding in rat brain membranes". Biological & Pharmaceutical Bulletin. 30 (12): 2328–2333. doi:10.1248/bpb.30.2328. PMID 18057721.
- ^ Arai Y, Toyoshima Y, Kinemuchi H (June 1986). "Studies of monoamine oxidase and semicarbazide-sensitive amine oxidase. II. Inhibition by alpha-methylated substrate-analogue monoamines, alpha-methyltryptamine, alpha-methylbenzylamine and two enantiomers of alpha-methylbenzylamine". Japanese Journal of Pharmacology. 41 (2): 191–197. doi:10.1254/jjp.41.191. PMID 3747266.
- ^ Greig ME, Walk RA, Gibbons AJ (October 1959). "The effect of three tryptamine derivatives on serotonin metabolism in vitro and in vivo". The Journal of Pharmacology and Experimental Therapeutics. 127: 110–115. PMID 13851725.
- ^ a b c Gey KF, Pletscher A (August 1962). "Effect of alpha-alkylated tryptamine derivatives on 5-hydroxytryptamine metabolism in vivo". British Journal of Pharmacology and Chemotherapy. 19 (1): 161–167. doi:10.1111/j.1476-5381.1962.tb01437.x. PMC 1482243. PMID 13898151.
- ^ Oeri HE (May 2021). "Beyond ecstasy: Alternative entactogens to 3,4-methylenedioxymethamphetamine with potential applications in psychotherapy". Journal of Psychopharmacology. 35 (5): 512–536. doi:10.1177/0269881120920420. PMC 8155739. PMID 32909493.
- ^ a b Glennon RA, Dukat MG (December 2023). "α-Ethyltryptamine: A Ratiocinatory Review of a Forgotten Antidepressant". ACS Pharmacology & Translational Science. 6 (12): 1780–1789. doi:10.1021/acsptsci.3c00139. PMC 10714429. PMID 38093842.
- ^ Huang XM, Johnson MP, Nichols DE (July 1991). "Reduction in brain serotonin markers by alpha-ethyltryptamine (Monase)". European Journal of Pharmacology. 200 (1): 187–190. doi:10.1016/0014-2999(91)90686-k. PMID 1722753.
- ^ Szara S (February 1961). "6-Hydroxylation: an important metabolic route for alpha-methyltryptamine". Experientia. 17 (2): 76–77. doi:10.1007/BF02171429. PMID 13774483. S2CID 27030395.
- ^ Kanamori T, Kuwayama K, Tsujikawa K, Miyaguchi H, Iwata YT, Inoue H (December 2008). "In vivo metabolism of alpha-methyltryptamine in rats: identification of urinary metabolites". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 38 (12): 1476–1486. doi:10.1080/00498250802491654. PMID 18982537. S2CID 20637936.
- ^ Sourkes TL (1991). "Alpha-methyltryptophan as a therapeutic agent". Prog Neuropsychopharmacol Biol Psychiatry. 15 (6): 935–938. doi:10.1016/0278-5846(91)90020-2. PMID 1763198.
- ^ Roberge AG, Missala K, Sourkes TL (March 1972). "Alpha-methyltryptophan: effects on synthesis and degradation of serotonin in the brain". Neuropharmacology. 11 (2): 197–209. doi:10.1016/0028-3908(72)90092-5. PMID 4260268.
- ^ Marsden CA, Curzon G (1977). "Effects of p-chlorophenylalanine and alpha-methyltryptophan on behaviour and brain 5-hydroxyindoles". Neuropharmacology. 16 (7–8): 489–494. doi:10.1016/0028-3908(77)90006-5. PMID 144245.
- ^ Anonymous. "Synthesis of alpha-Methyltryptamine (IT-290/AMT)". TheHive. Retrieved 7 June 2023.
- ^ Snyder HR, Katz L (December 1947). "The alkylation of aliphatic nitro compounds with gramine; a new synthesis of derivatives of tryptamine". Journal of the American Chemical Society. 69 (12): 3140–3142. doi:10.1021/ja01204a061. PMID 18919717.
- ^ Greig ME, Walk RA, Gibbons AJ (October 1959). "The effect of three tryptamine derivatives on serotonin metabolism in vitro and in vivo". The Journal of Pharmacology and Experimental Therapeutics. 127: 110–115. PMID 13851725.
- ^ Heinzelman RV, Anthony WC, Lyttle DA, Szmuszkovicz J (1960). "The Synthesis of α-Methyltryptophans and α-Alkyltryptamines". The Journal of Organic Chemistry. 25 (9): 1548–1558. doi:10.1021/jo01079a021. ISSN 0022-3263.
- ^ Murphree HB, Dippy RH, Jenney EH, Pfeiffer CC (1961). "Effects in normal man of alpha-methyltryptamine and alpha-ethyltryptamine". Clinical Pharmacology and Therapeutics. 2 (6): 722–726. doi:10.1002/cpt196126722. PMID 14477400.
- ^ Himwich HE (February 1961). "Experiments with alpha-methyltryptamine". Journal of Neuropsychiatry. 2 (Suppl 1): 136–140. PMID 13714418.
- ^ Vane JR, Collier HO, Corne SJ, Marley E, Bradley PB (September 1961). "Tryptamine receptors in the central nervous system". Nature. 191 (4793): 1068–1069. doi:10.1038/1911068a0. PMID 13780152.
- ^ Keller JG, Viguera C, Kundzins W (November 1962). "Studies on the toxicology of alpha-methyl- and alpha-ethyltryptamine acetates (Monase). II. Chronic studies". Toxicology and Applied Pharmacology. 4 (6): 697–709. doi:10.1016/0041-008x(62)90099-6. PMID 14031765.
- ^ Szara S, Hearst E, Putney F (1962). "Metabolism and behavioural action of psychotropic tryptamine homologues". International Journal of Neuropharmacology. 1 (1–3): 111–117. doi:10.1016/0028-3908(62)90015-1.
- ^ Mashkovskii MD, Trubitsyna TK (1963). "[Pharmacological properties of indopane (alpha-methyltryptamine HCl)]". Zhurnal Nevropatologii I Psikhiatrii imeni S.S. Korsakova (in Russian). 63: 72–79. PMID 13933321.
- ^ US Patent 3296072, Szmuszkovicz J, "Method of Treating Mental Depression", published 1967-01-03, assigned to Upjohn Co.
- ^ Elks J (2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer US. ISBN 978-1-4757-2085-3. Retrieved 7 September 2024.
- ^ Hollister LE, Prusmack JJ, Paulsen A, Rosenquist N (November 1960). "Comparison of three psychotropic drugs (psilocybin, JB-329, and IT-290) in volunteer subjects". The Journal of Nervous and Mental Disease. 131 (5): 428–434. doi:10.1097/00005053-196011000-00007. PMID 13715375.
- ^ Lessin AW, Long RF, Parkes MW (April 1966). "Conversion of indolylalkylhydroxylamines to stimulant amines in vivo". Biochemical Pharmacology. 15 (4): 481–487. doi:10.1016/0006-2952(66)90258-9. PMID 5954980.
- ^ Lessin AW, Long RF, Parkes MW (January 1967). "The central stimulant properties of some substituted indolylalkylamines and beta-carbolines and their activities as inhibitors of monoamine oxidase and the uptake of 5-hydroxytryptamine". British Journal of Pharmacology and Chemotherapy. 29 (1): 70–79. doi:10.1111/j.1476-5381.1967.tb01940.x. PMC 1557188. PMID 19108241.
- ^ Burningham RA, Arimura GK, Yunis AA (July 1966). "Effect of Monase and related compounds on uptake of 5-hydroxytryptamine by platelets". Proceedings of the Society for Experimental Biology and Medicine. 122 (3): 711–714. doi:10.3181/00379727-122-31233. PMID 5918937.
- ^ Offermeier J (1965). Serotonin and its derivatives: a study on structure-activity relations (Ph.D. thesis). Catholic University of Nijmegen. Retrieved 7 September 2024.
- ^ "("indopan"[title] OR "indopane"[title])". PubMed. U.S. National Library of Medicine. Retrieved 7 September 2024.
- ^ "5-MeO-AMT: Legal Status". Erowid Vault.
- ^ a b c d e f g Expert Committee on Drug Dependence Thirty-sixth Meeting (June 2014). "Alpha-methyltryptamine (AMT) Critical Review Report Agenda item 4.20" (PDF). Geneva: World Health Organization (WHO). Archived from the original (PDF) on 16 September 2018.
- ^ "CSDA". Archived from the original on 2011-09-28. Retrieved 2011-06-05.
- ^ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" [Notice on the issuance of the "Regulations on the Listing of Non-Medicinal Narcotic Drugs and Psychotropic Drugs"] (in Chinese). China Food and Drug Administration. 27 September 2015. Archived from the original on 1 October 2015. Retrieved 1 October 2015.
- ^ "Lääkeluettelon Aineet, Liite 1, Bilaga 1.3" [Substances of the drug list, Appendix 1. Attachment 1.3] (PDF). Finlex Data Bank. Finnish Ministry of Justice.
- ^ "Medicamentos de Uso Humano - Estupefacientes y Psicótropos" [Medicines for Human Use - Narcotics and Psychotropic Substances]. Agencia Española de Medicamentos y Productos Sanitarios [Spanish Agency for Medicines and Health Products]. Archived from the original on 2019-01-02. Retrieved 2019-01-01.
- ^ "Förordning om ändring i förordningen (1999:58) om förbud mot vissa hälsofarliga varor [Ordinance amending the Ordinance (1999:58) on the prohibition of certain goods hazardous to health]" (PDF), Lagen om förbud mot vissa hälsofarliga varor - SFS 2005:26 [The Act on the Prohibition of Certain Goods Hazardous to Health - SFS 2005:26] (in Swedish), February 2005, archived from the original (PDF) on 2013-09-29, retrieved 2013-10-07
- ^ "The Misuse of Drugs (Amendment No. 3) (England, Wales and Scotland) Regulations 2014". www.legislation.gov.uk.
- ^ ACMD (10 June 2014). "Update of the Generic Definition for Tryptamines" (PDF). UK Home Office. p. 12. Retrieved 10 June 2014.
- ^ "Alpha-methyltryptamine" (PDF). DEA Office of Diversion Control. April 2013. Archived (PDF) from the original on 2013-10-17. Retrieved 2013-10-10.